This is a modern-English version of Metabolic Adaptation to Climate and Distribution of the Raccoon Procyon Lotor and Other Procyonidae, originally written by Mahlke-Johnson, Kathleen P., Mugaas, John N., Seidensticker, John.
It has been thoroughly updated, including changes to sentence structure, words, spelling,
and grammar—to ensure clarity for contemporary readers, while preserving the original spirit and nuance. If
you click on a paragraph, you will see the original text that we modified, and you can toggle between the two versions.
Scroll to the bottom of this page and you will find a free ePUB download link for this book.

and Distribution of the Raccoon
Procyon lotor and Other Procyonidae
and Kathleen P. Mahlke-Johnson

SMITHSONIAN INSTITUTION PRESS
Washington, D.C.
1993
Mugaas, J. N., J. Seidensticker, and K. Mahlke-Johnson. Metabolic Adaptation to Climate and Distribution of the Raccoon Procyon lotor and Other Procyonidae. Smithsonian Contributions to Zoology, number 542, 34 pages, 8 figures, 12 tables, 1993.—Although the family Procyonidae is largely a Neotropical group, the North American raccoon, Procyon lotor, is more versatile in its use of climate, and it is found in nearly every habitat from Panama to 60°N in Canada. We hypothesized that most contemporary procyonids have remained in tropic and subtropic climates because they have retained the metabolic characteristics of their warm-adapted ancestors, whereas Procyon lotor evolved a different set of adaptations that have enabled it to generalize its use of habitats and climates. To test this hypothesis we compared Procyon lotor with several other procyonids (Bassariscus astutus, Nasua nasua, Nasua narica, Procyon cancrivorus, and Potos flavus) with respect to (1) basal metabolic rate (Ḣb), (2) minimum wet thermal conductance (Cmw), (3) diversity of diet (Dd), (4) intrinsic rate of natural increase (rmax), and, where possible, (5) capacity for evaporative cooling (Ec). We measured basal and thermoregulatory metabolism, evaporative water loss, and body temperature of both sexes of Procyon lotor from north central Virginia, in summer and winter. Metabolic data for other procyonids were from literature, as were dietary and reproductive data for all species.
Mugaas, J. N., J. Seidensticker, and K. Mahlke-Johnson. Metabolic Adaptation to Climate and Distribution of the Raccoon Procyon lotor and Other Procyonidae. Smithsonian Contributions to Zoology, number 542, 34 pages, 8 figures, 12 tables, 1993.—Although the Procyonidae family is mostly found in the Neotropics, the North American raccoon, Procyon lotor, is more adaptable to various climates and can be found in nearly every habitat from Panama to 60°N in Canada. We proposed that most modern procyonids have stayed in tropical and subtropical climates because they have kept the metabolic traits of their warm-adapted ancestors, while Procyon lotor developed a different set of adaptations that have allowed it to thrive in a variety of habitats and climates. To test this idea, we compared Procyon lotor with several other procyonids (Bassariscus astutus, Nasua nasua, Nasua narica, Procyon cancrivorus, and Potos flavus) regarding (1) basal metabolic rate (Ḣb), (2) minimum wet thermal conductance (Cmw), (3) dietary diversity (Dd), (4) intrinsic rate of natural increase (rmax), and, if possible, (5) capacity for evaporative cooling (Ec). We measured the basal and thermoregulatory metabolism, evaporative water loss, and body temperature of both male and female Procyon lotor from north central Virginia, during summer and winter. Metabolic data for other procyonids were obtained from literature, along with dietary and reproductive data for all species.
Procyon lotor differed from other procyonids in all five variables. (1) Procyon lotor's mass specific Ḣb (0.46 mL O2·g-1·h-1) was 1.45 to 1.86 times greater than values for other procyonids. (2) Because of its annual molt, Procyon lotor's Cmw was about 49% higher in summer than winter, 0.0256 and 0.0172 mL O2·g-1·h-1·°C-1, respectively. The ratio of measured to predicted Cmw for Procyon lotor in winter (1.15) was similar to values calculated for Potos flavus (1.02) and Procyon cancrivorus (1.25). Values for other procyonids were higher than this, but less than the value for Procyon lotor (1.76) in summer. On a mass specific basis, Bassariscus astutus had the lowest Cmw with a ratio of 0.85. (3) Procyon lotor utilized three times as many food categories as Procyon cancrivorus, Nasua nasua, and Bassariscus astutus; about two times as many as Nasua narica; and nine times as many as Potos flavus. (4) Intrinsic rate of natural increase correlated positively with Ḣb. Procyon lotor had the highest rmax (2.52 of expected) and Potos flavus the lowest (0.48 of expected). The other procyonids examined also had low Ḣb, but their rmax's were higher than predicted (1.11-1.32 of expected). Early age of first female reproduction, fairly large litter size, long life span, high-quality diet, and, in one case, female social organization all compensated for low Ḣb and elevated rmax. (5) Although data on the capacity for evaporative cooling were incomplete, this variable appeared to be best developed in Procyon lotor and Bassariscus astutus, the two species that have been most successful at including temperate climates in their distributions.
Procyon lotor was different from other procyonids in all five aspects. (1) Procyon lotor's mass-specific Hb (0.46 mL O2·g-1·h-1) was 1.45 to 1.86 times higher than values for other procyonids. (2) Due to its annual molt, Procyon lotor's Cmw was about 49% greater in summer than in winter, at 0.0256 and 0.0172 mL O2·g-1·h-1·°C-1, respectively. The ratio of measured to predicted Cmw for Procyon lotor in winter (1.15) was similar to the values calculated for Potos flavus (1.02) and Procyon cancrivorus (1.25). Values for other procyonids were higher than this, but still lower than the value for Procyon lotor (1.76) in summer. On a mass-specific basis, Bassariscus astutus had the lowest Cmw with a ratio of 0.85. (3) Procyon lotor used three times as many food categories as Procyon cancrivorus, Nasua nasua, and Bassariscus astutus; about twice as many as Nasua narica; and nine times as many as Potos flavus. (4) The intrinsic rate of natural increase correlated positively with Hb. Procyon lotor had the highest rmax (2.52 times the expected value) and Potos flavus the lowest (0.48 times the expected). The other procyonids studied also had low Hb, but their rmax's were higher than predicted (1.11-1.32 times the expected). An early age for first female reproduction, relatively large litter sizes, long lifespan, high-quality diet, and, in one case, female social organization all compensated for low Hb and increased rmax. (5) Although information on the capacity for evaporative cooling was incomplete, this characteristic seemed to be most developed in Procyon lotor and Bassariscus astutus, the two species that have been most successful at adapting to temperate climates in their ranges.
These five variables are functionally interrelated, and have co-evolved in each species to form a unique adaptive unit that regulates body temperature and energy balance throughout each annual cycle. The first four variables were converted into normalized dimensionless numbers, which were used to derive a composite score that represented each species' adaptive unit. Procyon lotor had the highest composite score (1.47) and Potos flavus the lowest (0.39). Scores for the other procyonids were intermediate to these extremes (0.64-0.79). There was a positive correlation between the number of climates a species occupies and the magnitude of its composite score. Linear regression of this relationship indicated that 89% of the variance in climatic distribution was attributed to the composite scores. Differences in metabolic adaptation, therefore, have played a role in delimiting climatic distribution of these species.
These five variables are closely connected and have evolved together in each species to create a unique adaptive unit that controls body temperature and energy balance throughout the year. The first four variables were turned into normalized dimensionless numbers, which were then used to calculate a composite score representing each species' adaptive unit. Procyon lotor had the highest composite score (1.47), while Potos flavus had the lowest (0.39). The scores for the other procyonids fell in between these two extremes (0.64-0.79). There was a positive correlation between the number of climates a species inhabits and the size of its composite score. A linear regression of this relationship showed that 89% of the variance in climatic distribution was due to the composite scores. Thus, differences in metabolic adaptation have contributed to defining the climatic range of these species.
It was clear that Procyon lotor differed from the other procyonids with respect to thermoregulatory ability, diet, and reproductive potential. These differences have enabled it to become a highly successful climate generalist, and its evolution of an Ḣb that is higher than the procyonid norm appears to be the cornerstone of its success.
It was clear that Procyon lotor was different from the other procyonids in terms of its ability to regulate body temperature, diet, and reproductive potential. These differences have allowed it to thrive as a successful climate generalist, and its evolution of an Ḣb that is higher than the typical procyonid seems to be the key to its success.
Official publication date is handstamped in a limited number of initial copies and is recorded in the Institution's annual report, Smithsonian Year. Series cover design: The coral Montastrea cavernosa (Linnaeus).
Release date is stamped by hand in a limited number of initial copies and is recorded in the Institution's annual report, Smithsonian Year. Series cover design: The coral Montastrea cavernosa (Linnaeus).
Library of Congress Cataloging-in-Publication Data
Mugaas, John N.
Metabolic adaptation to climate and distribution of the raccoon Procyon lotor and other Procyonidae / John N. Mugaas,
John Seidensticker, and Kathleen P. Mahlke-Johnson.
p. cm.—(Smithsonian contributions to zoology; no. 542)
Includes bibliographical references (p. )
1. Raccoons-Metabolism-Climatic factors. 2. Procyonidae-Metabolism-Climatic factors. 3. Raccoons-Geographical
distribution. 4. Procyonidae-Geographical distribution. I. Seidensticker, John. II. Mahlke-Johnson,
Kathleen. III. Title. IV. Series.
QL1.S54 no. 542 [QL737.C26] 591 s-dc20 [599.74´443´04542] 93-3119
![]() The paper used in this publication meets the minimum requirements of the American
National Standard for Permanence of Paper for Printed Library Materials z39.48—1984.
The paper used in this publication meets the minimum requirements of the American
National Standard for Permanence of Paper for Printed Library Materials z39.48—1984.
Library of Congress Cataloging-in-Publication Data
Mugaas, John N.
Metabolic adaptation to climate and distribution of the raccoon Procyon lotor and other Procyonidae / John N. Mugaas,
John Seidensticker, and Kathleen P. Mahlke-Johnson.
p. cm.—(Smithsonian contributions to zoology; no. 542)
Includes bibliographical references (p. )
1. Raccoons-Metabolism-Climatic factors. 2. Procyonidae-Metabolism-Climatic factors. 3. Raccoons-Geographical
distribution. 4. Procyonidae-Geographical distribution. I. Seidensticker, John. II. Mahlke-Johnson,
Kathleen. III. Title. IV. Series.
QL1.S54 no. 542 [QL737.C26] 591 s-dc20 [599.74´443´04542] 93-3119
![]() The paper used in this publication meets the minimum requirements of the American
National Standard for Permanence of Paper for Printed Library Materials z39.48—1984.
The paper used in this publication meets the minimum requirements of the American
National Standard for Permanence of Paper for Printed Library Materials z39.48—1984.
and Distribution of the Raccoon
Procyon lotor and Other Procyonidae
and Kathleen P. Mahlke-Johnson
The major carnivore radiations took place about 40 million years before present (MYBP) in the late Eocene and early Oligocene (Ewer, 1973:363; Wayne et al., 1989). Between 30 and 40 MYBP, a progenitor split into the ursid and procyonid lineages, which evolved into present-day bears, pandas, and raccoons (Wayne et al., 1989). The taxonomic relatedness of pandas to bears and raccoons has been tested extensively and a number of authors have summarized current thinking on the problem (Martin, 1989; Wayne et al., 1989; Wozencraft, 1989a, 1989b; Decker and Wozencraft, 1991). Davis (1964:322-327) and others (Leone and Wiens, 1956; Todd and Pressman, 1968; Sarich, 1976; O'Brien et al., 1985) place the giant panda, Ailuropoda melanoleuca, with the ursids. The taxonomic status of the red panda, Ailurus fulgens, appears to be less certain. Some current investigations align the red panda with bears (Segall, 1943; Todd and Pressman, 1968; Hunt, 1974; Ginsburg, 1982; Wozencraft, 1984:56-110; 1989a), whereas others place them intermediate to procyonids and bears (Wurster and Benirschke, 1968; Sarich, 1976; O'Brien et al., 1985), or in close relationship to the giant panda (Tagle et al., 1986).
The main radiations of carnivores happened around 40 million years ago (MYBP) in the late Eocene and early Oligocene (Ewer, 1973:363; Wayne et al., 1989). Between 30 and 40 MYBP, a common ancestor branched off into the bear and raccoon lineages, which evolved into the modern bears, pandas, and raccoons (Wayne et al., 1989). The taxonomic relationships of pandas with bears and raccoons have been extensively researched, and several authors have summarized the current understanding of this issue (Martin, 1989; Wayne et al., 1989; Wozencraft, 1989a, 1989b; Decker and Wozencraft, 1991). Davis (1964:322-327) and others (Leone and Wiens, 1956; Todd and Pressman, 1968; Sarich, 1976; O'Brien et al., 1985) classify the giant panda, Ailuropoda melanoleuca, within the bear family. The taxonomic classification of the red panda, Ailurus fulgens, seems to be less clear. Some recent studies group the red panda with bears (Segall, 1943; Todd and Pressman, 1968; Hunt, 1974; Ginsburg, 1982; Wozencraft, 1984:56-110; 1989a), while others place it somewhere between raccoons and bears (Wurster and Benirschke, 1968; Sarich, 1976; O'Brien et al., 1985), or closely related to the giant panda (Tagle et al., 1986).
The procyonid radiation took place in North America and produced forms that were mostly arboreal and omnivorous (Eisenberg, 1981:122; Martin, 1989). The center of this diversification occurred in Middle America (Baskin, 1982; Webb, 1985b) during the Miocene (Darlington, 1963:367; Webb, 1985b). Fossil procyonids from the late Miocene are represented in Florida, California, Texas, Nebraska, Kansas, and South Dakota (Baskin, 1982; Martin, 1989) and include such genera as Bassariscus, Arctonasua, Cyonasua, Paranasua, Nasua, and Procyon (Baskin, 1982; Webb, 1985b). During the Miocene procyonids underwent a modest radiation within tropical and subtropical climates of North America's central and middle latitudes. Cyonasua, which has close affinities to Arctonasua (Baskin, 1982), appears in tropical South America in the late Miocene and immigrated there either by rafting across the Bolivar Trough or by island-hopping through the Antilles archipelagoes (Marshall et al., 1982; Marshall, 1988). Thus, procyonids were found on both continents prior to formation of the Panamanian land bridge (Darlington, 1963:367, 395; Marshall et al., 1982; Marshall, 1988). Origins of Bassaricyon and Potos are obscure but probably occurred in tropical rainforests of Middle America (Baskin, 1982; Webb, 1985b). A subsequent Pleistocene dispersal carried several modern genera (Table 1) across the Panamanian land bridge into South America (Webb, 1985b). Bassariscus and Bassaricyon represent the most primitive genera in Procyoninae and Potosinae subfamilies, respectively (Table 1; Wozencraft, 1989a; Decker and Wozencraft, 1991).
The procyonid radiation occurred in North America and produced species that were mainly tree-dwelling and omnivorous (Eisenberg, 1981:122; Martin, 1989). The heart of this diversification took place in Central America (Baskin, 1982; Webb, 1985b) during the Miocene (Darlington, 1963:367; Webb, 1985b). Fossil procyonids from the late Miocene have been found in Florida, California, Texas, Nebraska, Kansas, and South Dakota (Baskin, 1982; Martin, 1989) and include genera like Bassariscus, Arctonasua, Cyonasua, Paranasua, Nasua, and Procyon (Baskin, 1982; Webb, 1985b). During the Miocene, procyonids experienced a slight radiation in the tropical and subtropical regions of North America's central and middle latitudes. Cyonasua, which is closely related to Arctonasua (Baskin, 1982), appears in tropical South America in the late Miocene and likely arrived there by rafting across the Bolivar Trough or by traveling through the Antilles archipelagoes (Marshall et al., 1982; Marshall, 1988). Therefore, procyonids were present on both continents before the formation of the Panamanian land bridge (Darlington, 1963:367, 395; Marshall et al., 1982; Marshall, 1988). The origins of Bassaricyon and Potos are unclear but probably took place in the tropical rainforests of Central America (Baskin, 1982; Webb, 1985b). A later Pleistocene dispersal brought several modern genera (Table 1) across the Panamanian land bridge into South America (Webb, 1985b). Bassariscus and Bassaricyon are the most primitive genera in the Procyoninae and Potosinae subfamilies, respectively (Table 1; Wozencraft, 1989a; Decker and Wozencraft, 1991).
In the early Tertiary, mid-latitudes of North America were much warmer than they are now, but not fully tropical, and temperate deciduous forests, associated with strongly seasonal climates, occurred only in the far north (Barghoorn, 1953; Colbert, 1953; Darlington, 1963:589, 590). Major climatic deteriorations, with their attendant cooling of northern continents, occurred during the Eo-Oligocene transition, in the middle Miocene, at the end of the Miocene, and at about 3 MYBP (late Pliocene). This last deterioration corresponds with closure of the Panamanian isthmus (Berggren, 1982; Webb, [Pg 2] 1985a). Climatic deterioration went on at an accelerating rate during the late Tertiary, with glacial conditions developing at the poles by the mid-Pliocene (Barghoorn, 1953). Therefore, throughout the Tertiary, as continents cooled, northern climate zones moved toward the tropics (Barghoorn, 1953; Colbert, 1953; Darlington, 1963:589, 590, 594, 595; Webb, 1985a).
In the early Tertiary, the mid-latitudes of North America were much warmer than they are today, but not fully tropical, and temperate deciduous forests that thrive in strongly seasonal climates existed only in the far north (Barghoorn, 1953; Colbert, 1953; Darlington, 1963:589, 590). Significant climatic downturns, leading to the cooling of northern continents, happened during the Eo-Oligocene transition, in the middle Miocene, at the end of the Miocene, and around 3 million years ago (late Pliocene). This last downturn coincided with the closure of the Panamanian isthmus (Berggren, 1982; Webb, [Pg 2] 1985a). The cooling trend continued to accelerate during the late Tertiary, with glacial conditions emerging at the poles by the mid-Pliocene (Barghoorn, 1953). Thus, throughout the Tertiary, as the continents cooled, northern climate zones shifted toward the tropics (Barghoorn, 1953; Colbert, 1953; Darlington, 1963:589, 590, 594, 595; Webb, 1985a).
Order Carnivora Bowdich, 1821
Suborder Caniformia Kretzoi, 1945
Family Procyonidae Gray, 1825
Subfamily Potosinae Trouessart, 1904
Genus Potos E. Geoffroy and G. Cuvier, 1795
P. flavus (S.A., C.A., M.)
Genus Bassaricyon Allen, 1876
B. alleni[A] (S.A.)
B. beddardi[A] (S.A.)
B. gabbii[A] (nS.A., C.A.)
B. lasius[A] (C.A.)
B. pauli[A] (C.A.)
Subfamily Procyoninae Gray, 1825
Genus Bassariscus Coues, 1887
B. astutus (M., wU.S.)
B. sumichrasti (C.A., M.)
Genus Nasua Storr, 1780
N. narica[B] (nS.A., C.A., M., swU.S.)
N. nasua[B] (S.A., sC.A.)
Genus Nasuella Hollister, 1915
N. olivacea (S.A.)
Genus Procyon Storr, 1780
P. cancrivorus (S.A., sC.A.)
P. gloveralleni[C] (Barbados)
P. insularis[C] (Maria Madre Is., Maria Magdalene Is.)
P. lotor[C] (C.A., M., U.S., sC.)
P. maynardi[C] (Bahamas, New Providence Is.)
P. minor[C] (Guadeloupe Is.)
P. pygmaeus[C] (M., Quintana Roo, Cozumel Is.)
Order Carnivores Bowdich, 1821
Suborder Caniformia Kretzoi, 1945
Family Procyonidae Gray, 1825
Subfamily Potosinae Trouessart, 1904
Genus Potos E. Geoffroy and G. Cuvier, 1795
P. flavus (S.A., C.A., M.)
Genus Bassaricyon Allen, 1876
B. alleni__A_TAG_PLACEHOLDER_0__ (S.A.)
B. beddardi__A_TAG_PLACEHOLDER_0__ (S.A.)
B. gabbii__A_TAG_PLACEHOLDER_0__ (nS.A., C.A.)
B. lasius__A_TAG_PLACEHOLDER_0__ (C.A.)
B. pauli__A_TAG_PLACEHOLDER_0__ (C.A.)
Subfamily Procyoninae Gray, 1825
Genus Bassariscus Coues, 1887
B. astutus (M., wU.S.)
B. sumichrasti (C.A., M.)
Genus Nasua Storr, 1780
N. narica__A_TAG_PLACEHOLDER_0__ (nS.A., C.A., M., swU.S.)
N. nasua__A_TAG_PLACEHOLDER_0__ (S.A., sC.A.)
Genus Nasuella Hollister, 1915
N. olivacea (S.A.)
Genus Procyon Storr, 1780
P. cancrivorus (S.A., sC.A.)
P. gloveralleni__A_TAG_PLACEHOLDER_0__ (Barbados)
P. insularis__A_TAG_PLACEHOLDER_0__ (Maria Madre Island, Maria Magdalene Island)
P. lotor__A_TAG_PLACEHOLDER_0__ (C.A., M., U.S., sC.)
P. maynardi__A_TAG_PLACEHOLDER_0__ (Bahamas, New Providence Island)
P. minor__A_TAG_PLACEHOLDER_0__ (Guadeloupe Island)
P. pygmaeus__A_TAG_PLACEHOLDER_0__ (M., Quintana Roo, Cozumel Island)
[B] These are considered conspecific in some current taxonomies (Kortlucke and Ramirez-Pulido, 1982); however, the scheme followed here maintains them as separate species (Decker, 1991).
[C] Several named forms of Procyon are a single species, Procyon lotor (Wozencraft, 1989a).
During the late Miocene, late Pliocene, and Pleistocene, the Bering land bridge between North America and Asia formed periodically, offering an avenue for dispersal between northern continents (Darlington, 1963:366; Webb, 1985a). However, by the late Tertiary, northern continents had cooled to the extent that climate, with its attendant sharply defined vegetative zones, became the major factor limiting dispersal by this route (Darlington, 1963:366; Webb, 1985a). Those Holarctic mammals that did cross the Bering land bridge in the late Tertiary were "cold-adapted" species associated with relatively cool, but not alpine, climates (Darlington, 1963:366; Ewer, 1973:369). Among carnivores this included some canids, ursids, mustelids, and felids (Darlington, 1963:393-395, 397; Webb, 1985a). Procyonids, however, did not cross the Bering land bridge into Asia, and Ewer (1973:369) ascribes this to their being an "essentially tropical group." Miocene radiation of procyonids occurred at a time when two of the four major climatic deteriorations (middle and late Miocene) were taking place (Webb, 1985a, 1985b). These deteriorations had the effect of cooling the middle latitudes to the extent that temperate forest forms began to appear in mid-latitude floras, along with a rapid influx of herbaceous plants (Barghoorn, 1953). The procyonid radiation did not penetrate beyond these climatically changing middle latitudes, which implies that these animals were "warm-adapted," and were, therefore, physiologically excluded from reaching the Bering land bridge. Today, three of the six genera and over half of the 18 species that comprise Procyonidae (Table 1; Wozencraft, 1989b) remain confined to tropical regions of North and South America (Hall and Kelson, 1959:878-897; Poglayen-Neuwall, 1975; Kortlucke and Ramirez-Pulido, 1982; Nowak and Paradiso, 1983:977-985).
During the late Miocene, late Pliocene, and Pleistocene, the Bering land bridge between North America and Asia formed at various times, providing a pathway for species to spread between the northern continents (Darlington, 1963:366; Webb, 1985a). However, by the late Tertiary, the northern continents had cooled to the point where climate, along with its clearly defined vegetation zones, became the main factor limiting movement along this route (Darlington, 1963:366; Webb, 1985a). The Holarctic mammals that crossed the Bering land bridge in the late Tertiary were species adapted to cooler, but not alpine, climates (Darlington, 1963:366; Ewer, 1973:369). This included some canids, ursids, mustelids, and felids among carnivores (Darlington, 1963:393-395, 397; Webb, 1985a). However, procyonids did not cross the Bering land bridge into Asia, and Ewer (1973:369) attributes this to them being "essentially tropical." The procyonid expansion took place during a time when two of the four major climatic deteriorations (middle and late Miocene) were happening (Webb, 1985a, 1985b). These deteriorations cooled the middle latitudes enough that temperate forest types started to show up in mid-latitude floras, along with a quick influx of herbaceous plants (Barghoorn, 1953). The procyonid radiation didn't extend beyond these changing middle latitudes, suggesting these animals were "warm-adapted" and were physiologically unable to reach the Bering land bridge. Today, three of the six genera and over half of the 18 species that make up the Procyonidae (Table 1; Wozencraft, 1989b) are still confined to tropical regions of North and South America (Hall and Kelson, 1959:878-897; Poglayen-Neuwall, 1975; Kortlucke and Ramirez-Pulido, 1982; Nowak and Paradiso, 1983:977-985).
McNab (1988a) contends that basal metabolism is a highly plastic character in evolution, and he has amply shown that ecologically uniform species are more apt to share common metabolic rates than taxonomically allied species from drastically different environments (McNab, 1984a, 1986a, 1986b, 1988a). Procyonids represent a taxonomically allied group that shared a common ecological situation for millions of years; consequently, members of this family might be expected to show some uniformity in their Ḣb. Basal and thermoregulatory metabolism of several procyonids have been measured: kinkajou, Potos flavus (Müller and Kulzer, 1977; McNab, 1978a; Müller and Rost, 1983), coatis, Nasua nasua (Chevillard-Hugot et al., 1980; Mugaas et al., in prep.), and Nasua narica (Scholander et al., 1950c; Mugaas et al., in prep.), ringtail, Bassariscus astutus (Chevalier, 1985), and crab-eating raccoon, Procyon cancrivorus (Scholander et al., 1950c). In general, these species have Ḣb's that are 40%-80% of the values predicted for them by the Kleiber (1961:206) equation. Lower than predicted Ḣb is viewed as an energy-saving adaptation for procyonids living in relatively stable tropical climates (Müller and Kulzer, 1977; Chevillard-Hugot et al., 1980; Müller and Rost, 1983). This implies that lower than predicted Ḣb is a general procyonid condition and that it represents a characteristic that evolved in response to the family's long association with tropical and subtropical forest environments.[Pg 3]
McNab (1988a) argues that basal metabolism is highly adaptable in evolution, and he has clearly demonstrated that ecologically similar species are more likely to share similar metabolic rates than related species from very different environments (McNab, 1984a, 1986a, 1986b, 1988a). Procyonids are a related group that shared a common ecological setting for millions of years; therefore, we might expect members of this family to exhibit some consistency in their Ḣb. The basal and thermoregulatory metabolism of several procyonids have been measured: kinkajou, Potos flavus (Müller and Kulzer, 1977; McNab, 1978a; Müller and Rost, 1983), coatis, Nasua nasua (Chevillard-Hugot et al., 1980; Mugaas et al., in prep.), and Nasua narica (Scholander et al., 1950c; Mugaas et al., in prep.), ringtail, Bassariscus astutus (Chevalier, 1985), and crab-eating raccoon, Procyon cancrivorus (Scholander et al., 1950c). Overall, these species have Ḣb's that are 40%-80% of the values expected from the Kleiber (1961:206) equation. A lower than expected Ḣb is seen as an energy-saving adaptation for procyonids living in relatively stable tropical climates (Müller and Kulzer, 1977; Chevillard-Hugot et al., 1980; Müller and Rost, 1983). This suggests that a below-average Ḣb is a common trait among procyonids and represents an adaptation developed in response to the family's long-term relationship with tropical and subtropical forest environments.[Pg 3]
Although most procyonids are found in only tropical to subtropical climates, the North American raccoon, Procyon lotor, (Figure 1) has a much broader distribution that extends from tropical Panama (8°N) to southern Canada. In Alberta, Canada, its range reaches the edge of the Hudsonian Life Zone at 60°N (for distribution maps see Hall and Kelson, 1959:878-897, and Poglayen-Neuwall, 1975). Range extensions and an increase in numbers have been noted in Canada and in parts of the United States since the 19th century (Lotze and Anderson, 1979; Kaufmann, 1982; Nowak and Paradiso, 1983:977-985). Thus, Procyon lotor is more complex ecologically than other procyonids, particularly when one takes into account its highly generalized food habits (Hamilton, 1936; Stuewer, 1943; Stains, 1956:39-51; Greenwood, 1981) and the wide range of habitat types (forest, prairie, desert, mountain, coastal marsh, freshwater marsh) and climates (tropical to north temperate) in which it is successful (Whitney and Underwood, 1952:1; Hall and Kelson, 1959:885; Lotze and Anderson, 1979; Kaufmann, 1982). On this basis it is clear that Procyon lotor has deviated from the typical procyonid portrait and has become the consummate generalist of the Procyonidae.
Although most procyonids are found only in tropical to subtropical climates, the North American raccoon, Procyon lotor, (Figure 1) has a much wider distribution that stretches from tropical Panama (8°N) to southern Canada. In Alberta, Canada, its range extends to the edge of the Hudsonian Life Zone at 60°N (for distribution maps see Hall and Kelson, 1959:878-897, and Poglayen-Neuwall, 1975). Range extensions and an increase in population numbers have been observed in Canada and parts of the United States since the 19th century (Lotze and Anderson, 1979; Kaufmann, 1982; Nowak and Paradiso, 1983:977-985). Thus, Procyon lotor is ecologically more complex than other procyonids, especially when considering its highly varied diet (Hamilton, 1936; Stuewer, 1943; Stains, 1956:39-51; Greenwood, 1981) and the wide array of habitat types (forest, prairie, desert, mountain, coastal marsh, freshwater marsh) and climates (tropical to north temperate) in which it thrives (Whitney and Underwood, 1952:1; Hall and Kelson, 1959:885; Lotze and Anderson, 1979; Kaufmann, 1982). Based on this, it's clear that Procyon lotor has strayed from the typical procyonid model and has become the ultimate generalist of the Procyonidae.

[Pg 4]Our general hypothesis was that whereas most contemporary procyonids have retained the metabolic characteristics of their warm-adapted ancestors, Procyon lotor possesses a different set of adaptations, which either evolved as characteristics unique to this species or were acquired from its ancestral stock. In either case, its unique adaptations have given Procyon lotor the physiological flexibility to generalize its use of habitats and climates and expand its geographic distribution to a much greater extent than other procyonids.
[Pg 4]Our general hypothesis was that while most modern procyonids have kept the metabolic traits of their warm-adapted ancestors, Procyon lotor has developed a different set of adaptations, which either evolved as unique traits of this species or were inherited from its ancestors. In both cases, these unique adaptations have provided Procyon lotor the physiological flexibility to adapt to a variety of habitats and climates, allowing it to expand its geographic range much more than other procyonids.
We tested our hypothesis by comparing Procyon lotor with several other procyonids (Bassariscus astutus, Nasua nasua, Nasua narica, Procyon cancrivorus, and Potos flavus) on the basis of their (1) basal metabolic rate (Ḣb), (2) minimum wet thermal conductance (Cmw), (3) diversity of diet (Dd), (4) intrinsic rate of natural increase (rmax), and, when data were available, (5) capacity for evaporative cooling (Ec). In a genetic sense each one of these variables is a complex adaptive characteristic, expression of which is determined by the interaction of several genes (Prosser, 1986:110-165). Experience has shown that a given species will express each one of these variables in a specific manner that is relevant to its mass, physiology, behavior, and environmental circumstance. Thus, different expressions of these variables may represent specific climatic adaptations (Prosser, 1986:16) that have been selected-for by evolutionary process. Because these variables are interrelated with respect to regulation of body temperature and energy balance, they have co-evolved in each species to form an adaptive unit. For each species, measured and calculated values for the first four variables were converted into dimensionless numbers and used to derive a composite score that represented its adaptive unit. Climatic distributions of these species were then compared relative to their composite scores.
We tested our hypothesis by comparing Procyon lotor with several other procyonids (Bassariscus astutus, Nasua nasua, Nasua narica, Procyon cancrivorus, and Potos flavus) based on their (1) basal metabolic rate (Ḣb), (2) minimum wet thermal conductance (Cmw), (3) diet diversity (Dd), (4) intrinsic rate of natural increase (rmax), and, whenever data were available, (5) capacity for evaporative cooling (Ec). Genetically speaking, each of these variables is a complex adaptive characteristic, the expression of which is shaped by the interaction of multiple genes (Prosser, 1986:110-165). Experience has shown that each species will express these variables in a specific way that relates to its mass, physiology, behavior, and environmental circumstances. Therefore, different expressions of these variables may signify specific climatic adaptations (Prosser, 1986:16) that have been favored by the process of evolution. Since these variables are interconnected regarding the regulation of body temperature and energy balance, they have co-evolved in each species to create an adaptive unit. For each species, the measured and calculated values for the first four variables were converted into dimensionless numbers and used to derive a composite score that represented its adaptive unit. The climatic distributions of these species were then compared in relation to their composite scores.
Basal metabolic rate represents the minimum energy required by an animal to maintain basic homeostasis (Lusk, 1917:141; Kleiber, 1932, 1961:251; Benedict, 1938; Brody, 1945:59; Robbins, 1983:105-111). For mammals, Ḣb appears to be determined by complex interactions between their body size (Kleiber, 1932, 1961:206; Benedict, 1938; Brody, 1945:368-374; Hemmingsen, 1960:15-36; McNab, 1983b; Calder, 1987), the climate in which they live (Scholander et al., 1950c; McNab and Morrison, 1963; Hulbert and Dawson, 1974; Shkolnik and Schmidt-Nielsen, 1976; McNab, 1979a; Vogel, 1980), their food habits (McNab, 1978a, 1978b, 1980a, 1983a, 1984a, 1986a, 1986b, 1988a, 1989), and their circadian period (Aschoff and Pohl, 1970; Prothero, 1984). Some species have higher mass-specific Ḣb than others, and this variation appears to be tied to ecological circumstances rather than taxonomic affinities (McNab, 1988a, 1989). Basal metabolic rate is important ecologically because it serves as a measure of a species' minimum "obligatory" energy requirement, and under many circumstances, it represents the largest energy demand associated with a daily energy budget (King, 1974:38-55; McNab, 1980a; Mugaas and King, 1981:37-40). Recently it also has been implicated as a permissive factor with respect to rmax of mammals (Hennemann, 1983; Lillegraven et al., 1987; Nicoll and Thompson, 1987; Thompson, 1987) via its direct effect on their rates of development and fecundity (McNab, 1980a, 1983a, 1986b; Hennemann, 1983; Schmitz and Lavigne, 1984; Glazier, 1985a, 1985b). The implication of this latter point is that those species with higher Ḣb's also have faster rates of development and greater fecundity and hence enjoy the competitive advantage of a higher rmax. Basal metabolism is, therefore, "a highly plastic character in the course of evolution" (McNab, 1988a:25) that has a profound influence on each species' life history.
Basal metabolic rate is the minimum energy an animal needs to maintain basic homeostasis (Lusk, 1917:141; Kleiber, 1932, 1961:251; Benedict, 1938; Brody, 1945:59; Robbins, 1983:105-111). For mammals, Ḣb seems to be shaped by complex interactions involving their body size (Kleiber, 1932, 1961:206; Benedict, 1938; Brody, 1945:368-374; Hemmingsen, 1960:15-36; McNab, 1983b; Calder, 1987), the climate where they live (Scholander et al., 1950c; McNab and Morrison, 1963; Hulbert and Dawson, 1974; Shkolnik and Schmidt-Nielsen, 1976; McNab, 1979a; Vogel, 1980), their eating habits (McNab, 1978a, 1978b, 1980a, 1983a, 1984a, 1986a, 1986b, 1988a, 1989), and their circadian rhythm (Aschoff and Pohl, 1970; Prothero, 1984). Some species have a higher mass-specific Ḣb than others, and this difference seems tied to ecological conditions rather than taxonomic relationships (McNab, 1988a, 1989). Basal metabolic rate is ecologically significant because it measures a species' minimum "obligatory" energy need, and in many cases, it represents the largest energy demand within a daily energy budget (King, 1974:38-55; McNab, 1980a; Mugaas and King, 1981:37-40). Recently, it has also been identified as a permissive factor regarding rmax in mammals (Hennemann, 1983; Lillegraven et al., 1987; Nicoll and Thompson, 1987; Thompson, 1987), influencing their rates of development and reproduction (McNab, 1980a, 1983a, 1986b; Hennemann, 1983; Schmitz and Lavigne, 1984; Glazier, 1985a, 1985b). This suggests that species with higher Ḣb's tend to develop faster and reproduce more, giving them a competitive edge with a higher rmax. Basal metabolism is, therefore, "a highly plastic character in the course of evolution" (McNab, 1988a:25) that significantly impacts each species' life history.
Whole-body resistance to passive heat transfer is equal to tissue resistance plus coat resistance. Within limits, these resistances can be altered; tissue resistance can be varied by changes in blood flow, whereas coat resistance can be changed by piloerection, molt, and behavior. When whole-body resistance is maximized (maximum tissue and coat resistances), passive heat transfer is minimized. The inverse of resistance is conductance; therefore, maximum whole-body resistance is the inverse of minimum thermal conductance (Cm). Minimum thermal conductance is readily derived from metabolic chamber data, and it is commonly used to describe an animal's capacity to minimize passive heat transfer. Minimum thermal conductance interacts with Ḣb and body mass to set the maximum temperature differential a mammal can maintain without increasing its basal level of heat production. The low temperature in this differential is the lower critical temperature (Tlc).
Whole-body resistance to passive heat transfer equals tissue resistance plus coat resistance. To some extent, these resistances can be adjusted; tissue resistance changes with blood flow, while coat resistance can be influenced by piloerection, molting, and behavior. When whole-body resistance is at its highest (maximum tissue and coat resistances), passive heat transfer is at its lowest. The inverse of resistance is conductance; therefore, maximum whole-body resistance is the inverse of minimum thermal conductance (Cm). Minimum thermal conductance is easily calculated from metabolic chamber data and is often used to describe an animal's ability to reduce passive heat transfer. Minimum thermal conductance interacts with Ḣb and body mass to determine the maximum temperature difference a mammal can maintain without raising its baseline heat production. The lower temperature in this difference is referred to as the lower critical temperature (Tlc).
Mass-specific Cm for mammals is negatively correlated with body mass (McNab and Morrison, 1963; Herreid and Kessel, 1967; McNab, 1970, 1979b; Bradley and Deavers, 1980; Aschoff, 1981), and for any given mass its magnitude is 52% higher during the active, rather than the inactive, phase of the daily cycle (Aschoff, 1981). However, some mammals have Cm's that are higher or lower than would be predicted for them on the basis of body mass and circadian phase. Seasonal [Pg 5] variation in Cm (higher values during summer than winter) has been reported for many northern mammals that experience large annual variations in air temperature (Scholander et al., 1950a; Irving et al., 1955; Hart, 1956, 1957; Irving, 1972:165). Some tropical mammals with very thin fur coats, and others with nearly hairless bodies, have high Cm's (McNab, 1984a), as do burrowing mammals (McNab, 1966, 1979b, 1984a) and the kit fox, Vulpes macrotis (Golightly and Ohmart, 1983). Some small mammals with low basal metabolic rates tend to have lower than predicted Cm's: small marsupials (McNab, 1978a), heteromyid rodents (McNab, 1979a), several ant eaters (McNab, 1984a), the arctic hare, Lepus arcticus (Wang et al., 1973), the ringtail, Bassariscus astutus (Chevalier, 1985), and the fennec, Fennecus zerda (Noll-Banholzer, 1979). Thus, in spite of its mass dependence, Cm also has been modified during the course of evolution by selective factors in the environment and by the animal's own metabolic characteristics.
Mass-specific Cm for mammals is negatively correlated with body mass (McNab and Morrison, 1963; Herreid and Kessel, 1967; McNab, 1970, 1979b; Bradley and Deavers, 1980; Aschoff, 1981), and for any given mass, its value is 52% higher during the active phase compared to the inactive phase of the daily cycle (Aschoff, 1981). However, some mammals have Cm values that are higher or lower than expected based on body mass and circadian phase. Seasonal variation in Cm (higher in summer than in winter) has been observed in many northern mammals that undergo significant annual temperature fluctuations (Scholander et al., 1950a; Irving et al., 1955; Hart, 1956, 1957; Irving, 1972:165). Some tropical mammals with very thin fur and others with almost hairless bodies have high Cm values (McNab, 1984a), as do burrowing mammals (McNab, 1966, 1979b, 1984a) and the kit fox, Vulpes macrotis (Golightly and Ohmart, 1983). Certain small mammals with low basal metabolic rates typically show lower Cm values than predicted: small marsupials (McNab, 1978a), heteromyid rodents (McNab, 1979a), several ants eaters (McNab, 1984a), the arctic hare, Lepus arcticus (Wang et al., 1973), the ringtail, Bassariscus astutus (Chevalier, 1985), and the fennec, Fennecus zerda (Noll-Banholzer, 1979). Therefore, despite its dependence on mass, Cm has also been shaped over time by environmental selective factors and the animal's own metabolic traits.
Latent heat loss occurs as a result of evaporation from the respiratory tract and through the skin, and except under conditions of heat stress, it "is a liability in thermal and osmotic homeostasis" (Calder and King, 1974:302). Ec, defined as the ratio of evaporative heat lost to metabolic heat produced, can be used to quantify thermoregulatory effectiveness of evaporative cooling and to make comparisons of heat tolerance between species. Thermoregulatory effectiveness of latent heat loss is not just a function of the rate of evaporative water loss but also of the rate of metabolic heat production (Lasiewski and Seymour, 1972). For example, a low metabolic rate minimizes endogenous heat load and thus conserves water, whereas the opposite is true of high metabolic rates (Lasiewski and Seymour, 1972). Some mammals that live in arid regions have evolved low metabolic rates and thus capitalize on this relationship to reduce their thermoregulatory water requirement (McNab and Morrison, 1963; McNab, 1966; MacMillen and Lee, 1970; Noll-Banholzer, 1979). What is evident, therefore, is that an animal's capacity for increasing latent heat loss must evolve together with its Ḣb and Cm in response to specific environmental demands.
Latent heat loss happens due to evaporation from the respiratory tract and the skin, and except in conditions of heat stress, it "is a liability in thermal and osmotic homeostasis" (Calder and King, 1974:302). Ec, which is defined as the ratio of evaporative heat lost to metabolic heat produced, can be used to measure the thermoregulatory effectiveness of evaporative cooling and to compare heat tolerance between species. The effectiveness of latent heat loss in thermoregulation is affected not only by the rate of evaporative water loss but also by the rate of metabolic heat production (Lasiewski and Seymour, 1972). For instance, a low metabolic rate reduces internal heat load and conserves water, while high metabolic rates do the opposite (Lasiewski and Seymour, 1972). Some mammals that live in dry areas have developed low metabolic rates to take advantage of this relationship, thereby reducing their need for thermoregulatory water (McNab and Morrison, 1963; McNab, 1966; MacMillen and Lee, 1970; Noll-Banholzer, 1979). Clearly, an animal's ability to increase latent heat loss must evolve alongside its Ḣb and Cm in response to specific environmental needs.
McNab (1986a, 1988a, 1989) demonstrated that, for mammals, departures of Ḣb from the Kleiber (1961:206) "norm" are highly correlated with diet and independent of phylogenetic relationships. McNab's analysis indicates that for mammals that feed on invertebrates, those species with body mass less than 100 g have Ḣb's that are equal to or greater than values predicted by the Kleiber equation, whereas those with body mass greater than 100 g have metabolic rates that are lower than predicted. Grazers, vertebrate eaters, nut eaters, and terrestrial frugivores also have Ḣb's that are equal to or greater than predicted, whereas insectivorous bats, arboreal folivores, arboreal frugivores, and terrestrial folivores all have rates that are lower than predicted. McNab (1986a) found animals with mixed diets harder to categorize, but in general he predicted that their Ḣb's would be related to (1) a food item that is constantly available throughout the year, (2) a food item that is most available during the worst conditions of the year, or (3) a mix of foods available during the worst time of the year. Although these correlations do not establish cause and effect between food habits and Ḣb, McNab's analysis does make it clear that the relationship between these variables has very real consequences for an animal's physiology, ecology, and evolution.
McNab (1986a, 1988a, 1989) showed that, for mammals, deviations of Ḣb from the Kleiber (1961:206) "norm" are strongly linked to diet and not influenced by phylogenetic relationships. McNab's analysis indicates that for mammals that eat invertebrates, species with a body mass of less than 100 g have Ḣb's that are equal to or greater than the values predicted by the Kleiber equation, while those with a body mass greater than 100 g have metabolic rates that are lower than expected. Grazers, vertebrate eaters, nut eaters, and terrestrial frugivores also have Ḣb's that meet or exceed predictions, whereas insect-eating bats, tree-dwelling leaf eaters, tree-dwelling fruit eaters, and ground-dwelling leaf eaters all have rates that are below predictions. McNab (1986a) found it more challenging to categorize animals with mixed diets, but generally, he predicted that their Ḣb's would relate to (1) a food source that is consistently available year-round, (2) a food source that is most abundant during the harshest conditions of the year, or (3) a combination of foods available during the worst time of the year. While these correlations do not prove a cause-and-effect relationship between dietary habits and Ḣb, McNab's analysis clearly indicates that the link between these variables has significant consequences for an animal's physiology, ecology, and evolution.
In this investigation we measured basal and thermoregulatory metabolism, evaporative water loss, and body temperature of raccoons from north central Virginia. Measurements were conducted on both sexes in summer and winter to determine how season and sex influenced these variables. We then compared the data for this widely distributed generalist with data from literature for its ecologically more restricted relatives. Dietary data for all species were taken from literature, as were reproductive data for calculation of rmax.
In this study, we measured the resting and heat-regulating metabolism, evaporative water loss, and body temperature of raccoons from north central Virginia. We took measurements from both males and females during summer and winter to see how the season and sex impacted these factors. We then compared our data for this broadly distributed generalist with information from literature on its more ecologically specialized relatives. Dietary information for all species was sourced from literature, as well as reproductive data for calculating rmax.
Our analysis demonstrated clear differences between Procyon lotor and other procyonids with respect to Ḣb, Cmw, Dd, and rmax. The composite score calculated from these variables for Procyon lotor was much higher than those derived for other species, and there was a positive correlation between the number of climates a species occupies and the magnitude of its composite score. Data on evaporative water loss, although not complete for all species, suggested that tropical and subtropical procyonids have less capacity for evaporative cooling than Procyon lotor or Bassariscus astutus. It was clear, therefore, that with respect to its thermal physiology, Procyon lotor differed markedly from other procyonids, and we contend that these differences have allowed this species to become a highly successful climate generalist and to expand its distribution into many different habitats and climates. Our analysis also suggested that the cornerstone of Procyon lotor's success as a climate generalist is its Ḣb, which is higher than the procyonid norm.
Our analysis showed clear differences between Procyon lotor and other procyonids regarding Ḣb, Cmw, Dd, and rmax. The composite score calculated from these variables for Procyon lotor was significantly higher than those for other species, and there was a positive correlation between the number of climates a species inhabits and the magnitude of its composite score. Data on evaporative water loss, although not complete for all species, indicated that tropical and subtropical procyonids have less ability for evaporative cooling than Procyon lotor or Bassariscus astutus. Therefore, it was evident that in terms of thermal physiology, Procyon lotor differed significantly from other procyonids, and we argue that these differences have enabled this species to become a highly successful climate generalist and to expand its range into various habitats and climates. Our analysis also suggested that the key to Procyon lotor's success as a climate generalist is its Ḣb, which is higher than the procyonid average.
The authors would like to thank John Eisenberg and Devra Kleiman for their support and encouragement throughout the study. This investigation was supported by research grants from the West Virginia School of Osteopathic Medicine (WVSOM), and Friends of the National Zoo (FONZ). Logistic support was provided by the National Zoological Park's Conservation and Research Center (CRC), and the departments of Mammalogy and Zoological Research. Our ability to [Pg 6] conduct physiological research at CRC was made possible by the thoughtful support and encouragement provided by Chris Wemmer. His excellent staff at CRC, especially Jack Williams, Junior Allison, and Red McDaniel, were very helpful in providing hospitality and logistical support to the senior author and his family during their various visits to the Center. The assistance of several people at the National Zoo also is gratefully acknowledged: Mitch Bush and Lyndsay Phillips not only provided veterinary support throughout the investigation, but also performed surgical procedures required to implant temperature-sensitive radio transmitters in several raccoons; Olav Oftedal made his laboratory available to us at various times and loaned us equipment to use at CRC; Miles Roberts and his staff provided care for our captive raccoons in the Department of Zoological Research during various parts of the investigation. Greg Sanders and Ken Halama, supported by FONZ assistantships, cared for our captive raccoons at CRC, provided assistance in the laboratory whenever needed, and were an invaluable source of aid. Their friendship and help is gratefully acknowledged. Ellen Broudy and Andy Meyer, supported by WVSOM and a student work study grant, respectively, provided assistance in the laboratory. David Brown, John Eisenberg, Mary Etta Hight, Brian McNab, Steve Thompson, and W. Chris Wozencraft critically reviewed various phases of the manuscript and provided many helpful suggestions. We deeply appreciate the work of Jean B. McConville, whose beneficial editorial suggestions helped us improve several early versions of the manuscript. We also gratefully acknowledge Diane M. Tyler, our editor at the Smithsonian Institution Press, whose expertise helped us mold the manuscript into its final form. Jill Mellon and Sriyanie Miththalapa, supported by FONZ traineeships, assisted in measuring the daily cycle of body temperature in raccoons. The Virginia Commission of Game and Inland Fisheries gave us permission to use wild-caught raccoons in this project.
The authors want to thank John Eisenberg and Devra Kleiman for their support and encouragement throughout the study. This research was funded by grants from the West Virginia School of Osteopathic Medicine (WVSOM) and Friends of the National Zoo (FONZ). Logistic support was provided by the National Zoological Park's Conservation and Research Center (CRC), along with the departments of Mammalogy and Zoological Research. Our ability to [Pg 6] conduct physiological research at CRC was made possible by the thoughtful support and encouragement from Chris Wemmer. His amazing team at CRC, especially Jack Williams, Junior Allison, and Red McDaniel, were incredibly helpful in providing hospitality and logistical support to the senior author and his family during their various visits to the Center. The assistance of several individuals at the National Zoo is also gratefully acknowledged: Mitch Bush and Lyndsay Phillips not only provided veterinary support throughout the investigation but also performed the surgical procedures necessary to implant temperature-sensitive radio transmitters in several raccoons; Olav Oftedal made his laboratory available to us at various times and lent us equipment to use at CRC; Miles Roberts and his staff cared for our captive raccoons in the Department of Zoological Research during different phases of the investigation. Greg Sanders and Ken Halama, supported by FONZ assistantships, took care of our captive raccoons at CRC, provided assistance in the lab whenever needed, and were an invaluable source of support. Their friendship and help are sincerely appreciated. Ellen Broudy and Andy Meyer, backed by WVSOM and a student work-study grant, respectively, assisted in the lab. David Brown, John Eisenberg, Mary Etta Hight, Brian McNab, Steve Thompson, and W. Chris Wozencraft reviewed various phases of the manuscript and offered many helpful suggestions. We are deeply grateful for the work of Jean B. McConville, whose valuable editorial suggestions helped us improve several early versions of the manuscript. We also sincerely thank Diane M. Tyler, our editor at the Smithsonian Institution Press, whose expertise helped us shape the manuscript into its final form. Jill Mellon and Sriyanie Miththalapa, supported by FONZ traineeships, helped measure the daily cycle of body temperature in raccoons. The Virginia Commission of Game and Inland Fisheries granted us permission to use wild-caught raccoons for this project.
Raccoons were caught from May 1980 through December 1984 on a trapping grid of 30 to 35 stations (one or two "live traps" per station) that covered about one-third of the National Zoological Park's Conservation and Research Center (CRC) near Front Royal, Virginia (Seidensticker et al., 1988; Hallett et al., 1991). Animals were trapped during 10 consecutive days each month, and in this five-year interval 407 raccoons were captured and marked with tattoos and ear tags. All captured animals were individualized with respect to age, reproductive status, physical condition, parasite load, and mass and body dimensions. These data characterized the structure and dynamics of the raccoon population at CRC and provided information on the annual cycle of fattening for raccoons in north central Virginia.
Raccoons were trapped from May 1980 to December 1984 on a grid of 30 to 35 stations (one or two "live traps" per station) that covered about one-third of the National Zoological Park's Conservation and Research Center (CRC) near Front Royal, Virginia (Seidensticker et al., 1988; Hallett et al., 1991). Animals were trapped over 10 consecutive days each month, and during this five-year period, 407 raccoons were captured and marked with tattoos and ear tags. All captured animals were recorded in terms of age, reproductive status, physical condition, parasite load, and weight and body measurements. This data helped characterize the structure and dynamics of the raccoon population at CRC and provided insights into the annual cycle of fattening for raccoons in north central Virginia.
Animals used for metabolic measurements were captured at CRC about 1.5 km south of the trapping grid and thus were genetically representative of the area. Six males were captured and measured during the summer of 1983. These animals were kept isolated for a week before being measured and were released later that summer at the site of their capture. The other seven animals used in our study were from the collection of the National Zoological Park and all of them had their origins at CRC.
Animals used for metabolic measurements were captured at CRC, located about 1.5 km south of the trapping grid, making them genetically representative of the area. Six males were captured and measured during the summer of 1983. These animals were kept isolated for a week before measurements were taken and were released later that summer at their capture site. The other seven animals used in our study were collected from the National Zoological Park, and all of them originally came from CRC.
Metabolic measurements, conducted at CRC, were carried out on eight males during July and August 1983, on four females and three males from November 1983 through March 1984, and on four females during June and July 1984.
Metabolic measurements at CRC were conducted on eight males from July to August 1983, on four females and three males from November 1983 to March 1984, and on four females during June and July 1984.
Raccoons were housed throughout the study such that they were constantly exposed to a natural cycle of temperature and photoperiod. Weather records for the Front Royal area indicate that average temperatures are around -0.5°C in January and 23.3°C in July (Crockett, 1972). Light:dark (L:D) periods for the latitude of CRC (48°55'N; United States Department of the Interior Geological Survey, 1972), calculated from duration of daylight tables (List, 1971:506-512), were 14.9:9.1 and 9.4:14.6 hours L:D for summer and winter solstices, respectively, and 12.2:11.8 hours L:D for vernal and autumnal equinoxes.
Raccoons were kept throughout the study in a way that allowed them to experience a natural cycle of temperature and light. Weather records for the Front Royal area show that the average temperatures are about -0.5°C in January and 23.3°C in July (Crockett, 1972). The light:dark (L:D) periods for the latitude of CRC (48°55'N; United States Department of the Interior Geological Survey, 1972), calculated from daylight duration tables (List, 1971:506-512), were 14.9:9.1 hours L:D for the summer solstice, 9.4:14.6 hours L:D for the winter solstice, and 12.2:11.8 hours L:D for the vernal and autumnal equinoxes.
Our animals were fed a measured amount of food daily, and they usually ate most of what was provided. Occasionally these animals would eat very little or none of their ration, and on some days they would eat all that was given to them. We fed them either feline diet (ground horse meat) or canned mackerel (Star-kist®[1]) along with high-protein dog chow (Purina®). When available, fresh fruit also was added to their diet. Water was always provided ad libitum.
Our animals were given a specific amount of food each day, and they usually ate most of it. Sometimes, these animals would eat very little or none of their portion, and on other days, they would consume everything we provided. We fed them either a feline diet (ground horse meat) or canned mackerel (Star-kist®[1]) along with high-protein dog food (Purina®). When we could, we also added fresh fruit to their meals. Water was always available for them to drink freely.
Measurements were conducted during the raccoons' daily inactive period (sunrise to sunset) in both summer and winter. Oxygen consumption was measured in a flow-through metabolism chamber at 5°C intervals from -10°C to 35°C. Animals were held at each temperature until the lowest rate of oxygen consumption had been obtained and maintained for at least 15 minutes. During each determination, oxygen consumption was monitored for 30 minutes to one hour beyond a suspected minimum value to see if an even lower reading could be obtained. Raccoons attained minimum levels of oxygen consumption more quickly at warm (>10°C) than at cold [Pg 7] temperatures. Depending on the temperature, therefore, each measurement took from two to five hours to complete. On days when two measurements could be completed, the second trial was always at a temperature 10°C warmer than the first.
Measurements were taken during the raccoons' daily resting period (sunrise to sunset) in both summer and winter. Oxygen consumption was recorded in a flow-through metabolism chamber at 5°C intervals, ranging from -10°C to 35°C. The animals were kept at each temperature until the lowest rate of oxygen consumption was reached and maintained for at least 15 minutes. During each measurement, oxygen consumption was monitored for 30 minutes to an hour beyond the suspected minimum value to check if an even lower reading could be recorded. Raccoons reached minimum levels of oxygen consumption more quickly at warmer temperatures (above 10°C) compared to colder temperatures. Depending on the temperature, each measurement therefore took between two to five hours to complete. On days when two measurements could be conducted, the second trial was always at a temperature 10°C higher than the first.
The metabolism chamber was constructed from galvanized sheet metal (77.5 × 45.5 × 51.0 cm = 180 liters) and was painted black inside. Within the chamber, the animal was held in a cage (71 × 39 × 33 cm) constructed from turkey wire that also was painted black. This cage prevented the raccoons from coming into contact with the walls of the chamber, yet it was large enough to allow them to stand and freely move about. The bottom of the cage was 11 cm above the chamber floor, which was covered to a depth of one cm with mineral oil to trap urine and feces.
The metabolism chamber was made from galvanized sheet metal (77.5 × 45.5 × 51.0 cm = 180 liters) and painted black on the inside. Inside the chamber, the animal was placed in a cage (71 × 39 × 33 cm) made from turkey wire, which was also painted black. This cage kept the raccoons from touching the chamber walls but was roomy enough for them to stand and move around freely. The bottom of the cage was 11 cm above the chamber floor, which was covered with one cm of mineral oil to collect urine and feces.
During measurements, the metabolism chamber was placed in a controlled-temperature cabinet (modified Montgomery Ward model 8969 freezer). Air temperature (Ta) in the metabolism chamber was regulated with a Yellow Springs Instrument model 74 temperature controller. Ta was controlled to ± 1.0°C at temperatures below freezing, and to ± 0.5°C at temperatures above freezing. The chamber air and wall temperatures were recorded continuously (Linseis model LS-64 recorder) during each experiment, and, except during temperature changes, they were always within 0.5°C of each other.
During the measurements, the metabolism chamber was placed in a temperature-controlled cabinet (modified Montgomery Ward model 8969 freezer). The air temperature (Ta) in the metabolism chamber was regulated using a Yellow Springs Instrument model 74 temperature controller. Ta was kept within ± 1.0°C at temperatures below freezing and ± 0.5°C at temperatures above freezing. The air and wall temperatures in the chamber were recorded continuously (Linseis model LS-64 recorder) during each experiment, and except during temperature changes, they were always within 0.5°C of each other.
Columns of Drierite® and Ascarite® removed water vapor and carbon dioxide, respectively, from air entering and leaving the chamber. Dry carbon-dioxide-free room air was pumped into the chamber (Gilman model 13152 pressure/vacuum pump) at a rate of 3.0 L/min (Gilmont model K3203-20 flow meter). Downstream from the chemical absorbents, an aliquot (0.1 L/min) of dry carbon-dioxide-free air was drawn off the chamber exhaust line and analyzed for oxygen content (Applied Electrochemistry model S-3A oxygen analyzer, model 22M analysis cell, and model R-1 flow control). All gas values were corrected to standard temperature and pressure for dry gas. Oxygen consumption was calculated from the difference in oxygen content between inlet and outlet air using Eq. 8 of Depocas and Hart (1957).
Columns of Drierite® and Ascarite® removed water vapor and carbon dioxide from the air entering and leaving the chamber. Dry, carbon-dioxide-free room air was pumped into the chamber using a Gilman model 13152 pressure/vacuum pump at a rate of 3.0 L/min, measured with a Gilmont model K3203-20 flow meter. Downstream from the chemical absorbents, an aliquot (0.1 L/min) of dry, carbon-dioxide-free air was taken from the chamber exhaust line and analyzed for oxygen content using an Applied Electrochemistry model S-3A oxygen analyzer, model 22M analysis cell, and model R-1 flow control. All gas values were corrected to standard temperature and pressure for dry gas. Oxygen consumption was calculated from the difference in oxygen content between inlet and outlet air using Eq. 8 of Depocas and Hart (1957).
Each raccoon was fasted for at least 12 hours before oxygen consumption measurements began. At the start and end of each metabolic trial the animal was weighed to the nearest 10 g (Doctors Infant Scale, Detecto Scales, Inc., Brooklyn, N.Y., U.S.A.). The body mass used in calculating minimum oxygen consumption and evaporative water loss was estimated from timed extrapolations of the difference between starting and ending weights, and the time at which these variables were measured.
Each raccoon was deprived of food for at least 12 hours before we started measuring their oxygen consumption. At the beginning and end of each metabolic trial, the animal was weighed to the nearest 10 g (Doctors Infant Scale, Detecto Scales, Inc., Brooklyn, N.Y., U.S.A.). The body mass used to calculate minimum oxygen consumption and evaporative water loss was estimated from timed extrapolations of the difference between the starting and ending weights, along with the timing of when these measurements were taken.
During metabolic measurements at temperatures above
freezing, evaporative water loss was determined gravimetrically.
Upstream from the chemical columns, an aliquot of air
(0.1 L/min) was drawn off the exhaust line and diverted for a
timed interval through a series of preweighed (0.1 mg) ![]() -tubes
containing Drierite®. The aliquot then passed through a second
series of
-tubes
containing Drierite®. The aliquot then passed through a second
series of ![]() -tubes containing Ascarite® before entering the
oxygen analysis system. Evaporative water loss was calculated
using Eq. 1
-tubes containing Ascarite® before entering the
oxygen analysis system. Evaporative water loss was calculated
using Eq. 1
During metabolic measurements at temperatures above freezing, evaporative water loss was determined by weight. Upstream from the chemical columns, a sample of air (0.1 L/min) was taken from the exhaust line and redirected for a set amount of time through a series of preweighed (0.1 mg) ![]() -tubes containing Drierite®. The sample then flowed through a second set of
-tubes containing Drierite®. The sample then flowed through a second set of ![]() -tubes containing Ascarite® before entering the oxygen analysis system. Evaporative water loss was calculated using Eq. 1
-tubes containing Ascarite® before entering the oxygen analysis system. Evaporative water loss was calculated using Eq. 1
| Ė = (mw·.Ve)/(.Va·t·m) | Eq. 1 |
where Ė is evaporative water loss (mg·g-1·h-1), mw is mass of
water collected (mg), .Ve is rate of air flow into the chamber (3.0
L/min), .Va is the rate of air flow through the ![]() -tubes (0.1 L/min),
t is length of the timed interval (h), and m is the estimated mass
of the raccoon at the time of sampling (g).
-tubes (0.1 L/min),
t is length of the timed interval (h), and m is the estimated mass
of the raccoon at the time of sampling (g).
where Ė is the evaporative water loss (mg·g-1·h-1), mw is the mass of water collected (mg), .Ve is the rate of air flow into the chamber (3.0 L/min), .Va is the rate of air flow through the ![]() -tubes (0.1 L/min), t is the length of the timed interval (h), and m is the estimated mass of the raccoon at the time of sampling (g).
-tubes (0.1 L/min), t is the length of the timed interval (h), and m is the estimated mass of the raccoon at the time of sampling (g).
Veterinarians at the National Zoological Park surgically implanted calibrated temperature-sensitive radio transmitters (Telonics, Inc., Mesa, AZ, U.S.A.) into abdominal cavities of two female and two male raccoons. Transmitter pulse periods were monitored with a digital processor (Telonics TDP-2) coupled to a receiver (Telonics TR-2-164/166). During some metabolic measurements, body temperatures of these animals were recorded to the nearest 0.1°C at 30-minute intervals. The daily cycle of body temperature of these raccoons also was measured once a month.
Veterinarians at the National Zoological Park surgically implanted calibrated, temperature-sensitive radio transmitters (Telonics, Inc., Mesa, AZ, U.S.A.) into the abdominal cavities of two female and two male raccoons. The transmitter pulse periods were monitored using a digital processor (Telonics TDP-2) connected to a receiver (Telonics TR-2-164/166). During some metabolic measurements, the body temperatures of these animals were recorded to the nearest 0.1°C at 30-minute intervals. The daily cycle of body temperature for these raccoons was also measured once a month.
At the conclusion of these experiments, the accuracy of our calorimetry apparatus was tested by burning an ethanol lamp in the metabolism chamber. During these tests a CO2 analyzer was incorporated into the system (Beckman, LB-2). Results demonstrated that we measured 84% of the oxygen consumed by the lamp as well as 84% of the water and CO2 it produced; standard deviation = ± 2.6, ± 5.0, and ± 3.6, respectively (n = 27). Average respiratory quotient (RQ) calculated from these data was O.657 ± 0.008 (n = 27), which is 99.5% of that predicted (0.66). McNab (1988b) reports that the accuracy of open-flow indirect calorimetry systems, such as ours, depends on the rate of air flow through the animal chamber. If flow rates are too low, there is inadequate mixing of air within the chamber, and the rate of oxygen consumption, as calculated from the difference in oxygen content of air flowing into and out of the chamber (Depocas and Hart, 1957), is underestimated. At some critical rate of air flow, which is unique to each combination of chamber and animal, this situation changes such that measured rates of oxygen consumption become independent of any further increase in flow rate (McNab, 1988b). In recent tests of our system, where we burned the ethanol lamp at a variety of chamber flow rates, the efficiency [Pg 8] of measurement increased linearly as flow rate increased, and the critical rate of air flow was about 6.7 L/min. This appeared to explain why a flow rate of 3.0 L/min underestimated oxygen consumption of the ethanol lamp.
At the end of these experiments, we tested the accuracy of our calorimetry equipment by burning an ethanol lamp in the metabolism chamber. During these tests, we added a CO2 analyzer to the system (Beckman, LB-2). The results showed that we accurately measured 84% of the oxygen consumed by the lamp, as well as 84% of the water and CO2 it produced; standard deviations were ± 2.6, ± 5.0, and ± 3.6, respectively (n = 27). The average respiratory quotient (RQ) calculated from this data was 0.657 ± 0.008 (n = 27), which is 99.5% of the predicted value (0.66). McNab (1988b) states that the accuracy of open-flow indirect calorimetry systems, like ours, relies on the air flow rate through the animal chamber. If the flow rates are too low, there isn’t enough mixing of air in the chamber, leading to an underestimation of the rate of oxygen consumption, as calculated from the difference in oxygen content of the air flowing in and out of the chamber (Depocas and Hart, 1957). At a certain critical air flow rate, which is unique to each combination of chamber and animal, this situation shifts so that the measured rates of oxygen consumption no longer depend on any further increase in flow rate (McNab, 1988b). In recent tests of our system, while burning the ethanol lamp at different chamber flow rates, the efficiency of measurement increased linearly with flow rate, and the critical air flow rate was about 6.7 L/min. This seemed to explain why a flow rate of 3.0 L/min underestimated the oxygen consumption of the ethanol lamp.
Our earlier tests of the efficiency of our system indicated that although we underestimated actual oxygen consumption of the ethanol lamp, we did so with a fair degree of precision; probably because flow rates were closely controlled. During our metabolic measurements, chamber flow rates also were closely controlled at 3.0 L/min, and we believe, therefore, that these measurements also were carried out with a high degree of precision. Consequently, all measured values of oxygen consumption and water production were considered to be 84% of their actual value and were adjusted to 100% before being included in this report.
Our earlier tests of how well our system works showed that even though we underestimated the actual oxygen usage of the ethanol lamp, we did it quite accurately; probably because we kept the flow rates tightly controlled. During our metabolic measurements, we also maintained chamber flow rates at 3.0 L/min, so we believe these measurements were conducted with a high level of accuracy as well. As a result, all recorded values for oxygen consumption and water production were deemed to be 84% of their actual value and were adjusted to 100% before being included in this report.
The calibration of all temperature-sensitive radio transmitters drifted over time. Transmitters were calibrated before they were surgically implanted and again after they were removed from the animals. Although the drift of each transmitter was unique, it was also linear (S. Tomkiewicz, Telonics, Inc., pers. com.). All body temperature measurements were corrected from timed extrapolations of the difference between starting and ending calibrations.
The calibration of all temperature-sensitive radio transmitters changed over time. Transmitters were calibrated before they were surgically implanted and again after they were removed from the animals. While each transmitter’s drift was unique, it was also linear (S. Tomkiewicz, Telonics, Inc., pers. com.). All body temperature measurements were adjusted based on timed extrapolations of the difference between the starting and ending calibrations.
Values of oxygen consumption, evaporative water loss, and body temperature were plotted as a function of chamber air temperature. Linear regressions of oxygen consumption at temperatures below the thermoneutral zone (Tn), and evaporative water loss at temperatures above freezing, were determined with the SAS (1982) GLM procedure. Lower critical temperature (Tlc) was determined graphically from intersection of the line representing Ḣb and the regression line representing oxygen consumption below Tn. Slopes and intercepts of regression lines, as well as other mean values, were compared with t-tests (Statistical Analysis System, 1982; Ott, 1984:138-175). Unless indicated otherwise, data are expressed as mean ± standard deviation (s.d.).
Values of oxygen consumption, evaporative water loss, and body temperature were plotted against chamber air temperature. Linear regressions of oxygen consumption at temperatures below the thermoneutral zone (Tn) and evaporative water loss at temperatures above freezing were calculated using the SAS (1982) GLM procedure. The lower critical temperature (Tlc) was determined graphically from the intersection of the line representing Ḣb and the regression line for oxygen consumption below Tn. Slopes and intercepts of the regression lines, along with other mean values, were compared using t-tests (Statistical Analysis System, 1982; Ott, 1984:138-175). Unless stated otherwise, data are presented as mean ± standard deviation (s.d.).
We employed the method first described by Cole (1954) to calculate rmax:
We used the method first described by Cole (1954) to calculate rmax:
| 1 = e-rmax + b·e-rmax(a) - b·e-rmax(n+1) | Eq. 2 |
where a is potential age of females first producing young, b is potential annual birth rate of female young, and n is potential age of females producing their final young. After life-history data were substituted into Eq. 2, rmax was determined by trial and error substitution (Hennemann, 1983).
where a is the potential age of females when they first have offspring, b is the potential yearly birth rate of female offspring, and n is the potential age of females when they have their last offspring. After inserting life-history data into Eq. 2, rmax was figured out through trial and error (Hennemann, 1983).
Because rmax represents the genetically fixed, physiologically determined maximum possible rate of increase, data on earliest possible age of female reproduction, highest possible birth rate of female young, and longest possible female reproductive life span were used for a, b, and n, respectively. Calculated values, therefore, represent physiologically possible, not ecologically possible, intrinsic rates of increase (Hennemann, 1983, 1984; Hayssen, 1984; McNab, 1984b). Values of n were derived from longevity records for captive animals, and as these were all large values of similar duration (14-16 years), they had very little effect on rmax. All species considered have one litter per year, and because their sex ratios at birth are about 50:50, variation in b was due to differences in litter size. Therefore, age of first reproduction and litter size had the greatest effect on rmax. Intrinsic rate of increase scales to body mass (Fenchel, 1974), and we removed this effect by comparing each calculated rmax with the value expected (rmaxe) on the basis of body mass (Hennemann, 1983).
Because rmax represents the genetically fixed, physiologically determined maximum possible rate of increase, data on the earliest possible age of female reproduction, the highest possible birth rate of female young, and the longest possible female reproductive lifespan were used for a, b, and n, respectively. The calculated values, therefore, represent physiologically possible, not ecologically possible, intrinsic rates of increase (Hennemann, 1983, 1984; Hayssen, 1984; McNab, 1984b). Values of n were derived from longevity records for captive animals, and since these were all large values of similar duration (14-16 years), they had very little effect on rmax. All species considered have one litter per year, and because their sex ratios at birth are about 50:50, variation in b was due to differences in litter size. Therefore, the age of first reproduction and litter size had the greatest effect on rmax. The intrinsic rate of increase scales to body mass (Fenchel, 1974), and we removed this effect by comparing each calculated rmax with the value expected (rmaxe) based on body mass (Hennemann, 1983).
Dimensionless numbers for each of the four variables used in calculating composite scores were derived as follows. Ratios of measured to predicted values were used for basal metabolism (Hbr) and minimum wet thermal conductance (Cmwr). Thermoregulatory ability at low temperatures is closely related to the ratio Hbr/Cmwr (McNab, 1966). This ratio was used, therefore, to gauge each species' cold tolerance. For Dd we used the ratio of food categories actually used by a species to the total number of food categories taken by all species tested (Ddr). The ratio of calculated to expected intrinsic rates of natural increase was used to derive rmaxr. Composite scores were calculated as
Dimensionless numbers for each of the four variables used to calculate composite scores were derived as follows. Ratios of measured to predicted values were used for basal metabolism (Hbr) and minimum wet thermal conductance (Cmwr). Thermoregulatory ability at low temperatures is closely related to the ratio Hbr/Cmwr (McNab, 1966). This ratio was used to assess each species' cold tolerance. For Dd, we used the ratio of food categories actually consumed by a species to the total number of food categories used by all species tested (Ddr). The ratio of calculated to expected intrinsic rates of natural increase was used to derive rmaxr. Composite scores were calculated as
| Composite score = [(Hbr/Cmwr) + Ddr + rmaxr]/3 | Eq. 3 |
The correlation between number of climates these species occupy and their composite scores was tested by linear regression.
The relationship between the number of climates these species inhabit and their overall scores was analyzed using linear regression.
According to monthly live-trapping records, the body mass of free-ranging female raccoons increased from 3.6 ± 0.6 kg during summer to 5.6 ± 0.8 kg in early winter, and the mass of free-ranging males increased from 4.0 ± 0.5 to 6.7 ± 0.9 kg during the same interval. These seasonal changes in body mass were due to fluctuations in the amount of body fat and represent a mechanism for storing energy during fall for use in winter. In summer, captive and trapped male and captive female raccoons had the same body mass (4.73 ± 0.61, 4.41 ± 0.70, and 4.67 [Pg 9] ± 0.88 kg, respectively, Table 2). Mass of captive females did not change between seasons, whereas captive males were heavier in winter than summer (p<0.005; Table 2). This seasonal change in mass of our captive males was of a much smaller magnitude (0.6 kg) than that observed for wild males (2.7 kg). During winter, captive males (5.34 ± 1.39 kg) were heavier than captive females (4.49 ± 0.98 kg; p<0.005; Table 2). Thus, our captive animals maintained a body mass throughout the year that was intermediate to the range of values found for wild raccoons in the same area.
According to monthly live-trapping records, the body weight of free-ranging female raccoons went up from 3.6 ± 0.6 kg in the summer to 5.6 ± 0.8 kg in early winter, while the weight of free-ranging males rose from 4.0 ± 0.5 to 6.7 ± 0.9 kg during the same period. These seasonal shifts in body weight were linked to changes in body fat and represent a way to store energy in the fall for winter use. In summer, both captive and trapped male raccoons, as well as captive female raccoons, had similar body weights (4.73 ± 0.61, 4.41 ± 0.70, and 4.67 ± 0.88 kg, respectively, Table 2). The weight of captive females stayed the same across seasons, while captive males were heavier in winter than in summer (p<0.005; Table 2). This seasonal weight change in our captive males was much smaller (0.6 kg) compared to what was seen in wild males (2.7 kg). During winter, captive males (5.34 ± 1.39 kg) weighed more than captive females (4.49 ± 0.98 kg; p<0.005; Table 2). Therefore, our captive animals maintained a body weight throughout the year that was in between the values found for wild raccoons in the same area.
| Season and sex | Body mass, ± s.d., | (n) | Basal metabolism, ± s.d., | (n) |
|---|---|---|---|---|
| Summer | ||||
| Trapped male | 4.41 ± 0.70 | (52) | 780 ± 112 | (20) |
| Captive male | 4.73 ± 0.61 | (22) | 680 ± 102 | (8) |
| Captive female | 4.67 ± 0.88 | (41) | 618 ± 92 | (13) |
| Winter | ||||
| Captive male | 5.34 ± 1.39 | (31) | 704 ± 81 | (19) |
| Captive female | 4.49 ± 0.98 | (42) | 667 ± 139 | (25) |
Within thermoneutrality, Ḣb (mL O2·g-1·h-1) was 0.54 ± 0.09 for trapped males in summer, 0.46 ± 0.07 for captive males in summer, 0.42 ± 0.07 for captive females in summer, 0.47 ± 0.06 for captive males in winter, and 0.46 ± 0.10 for captive females in winter (Figures 2, 3). Ratios of these measured values to those predicted by the Kleiber (1932, 1961:206) equation are 1.28, 1.12, 1.02, 1.17, and 1.09, respectively. To minimize the effect of body size (Mellen, 1963) and to facilitate comparisons between sexes and seasons and between captive and trapped animals, basal metabolism also was calculated as a function of metabolic body size (mL O2·kg-0.75·h-1; Table 2). Based on this analysis, trapped summer males had a higher basal metabolism than captive males (p<0.025) or females (p<0.005) in either season (Table 2). There was no difference in basal metabolism between captive males and females in either summer or winter, and there was no seasonal difference in their basal metabolic rates (Table 2).
Within the thermoneutral zone, Ḣb (mL O2·g-1·h-1) was 0.54 ± 0.09 for trapped males in summer, 0.46 ± 0.07 for captive males in summer, 0.42 ± 0.07 for captive females in summer, 0.47 ± 0.06 for captive males in winter, and 0.46 ± 0.10 for captive females in winter (Figures 2, 3). The ratios of these measured values to those predicted by the Kleiber (1932, 1961:206) equation are 1.28, 1.12, 1.02, 1.17, and 1.09, respectively. To reduce the impact of body size (Mellen, 1963) and to enable comparisons between genders and seasons, as well as between captive and wild animals, basal metabolism was also calculated based on metabolic body size (mL O2·kg-0.75·h-1; Table 2). This analysis showed that trapped summer males had a higher basal metabolism than captive males (p<0.025) or females (p<0.005) in either season (Table 2). There was no difference in basal metabolism between captive males and females during either summer or winter, and there was no seasonal difference in their basal metabolic rates (Table 2).
Minimum wet and dry thermal conductances were calculated using Eqs. 4 and 5
Minimum wet and dry thermal conductances were calculated using Eqs. 4 and 5
| Cmw = Ḣr / (Tb - Ta) | Eq. 4 |
| Cmd = (Ḣr - Ėeq) / (Tb - Ta) | Eq. 5 |
where Cmw is wet and Cmd is dry conductance (mL O2·g-1·h-1·°C-1); Ḣr is the lowest resting metabolic rate measured at each temperature (mL O2·g-1·h-1); Ėeq is oxygen equivalent for heat lost by evaporation [Ėeq = mL O2·g-1·h-1 = Ė·λ/γ, where Ė is evaporative water loss (mg·g-1·h-1), λ is heat of vaporization for water (2.43 J/mg), and γ is heat equivalent for oxygen (20.097 J/mL)]; Tb is body temperature (°C); and Ta is chamber air temperature (°C). Only data from animals equipped with temperature-sensitive radio transmitters were used for these calculations.
where Cmw is wet conductance and Cmd is dry conductance (mL O2·g-1·h-1·°C-1); Ḣr is the lowest resting metabolic rate recorded at each temperature (mL O2·g-1·h-1); Ėeq is the oxygen equivalent for heat lost through evaporation [Ėeq = mL O2·g-1·h-1 = Ė·λ/γ, where Ė is evaporative water loss (mg·g-1·h-1), λ is the heat of vaporization for water (2.43 J/mg), and γ is the heat equivalent for oxygen (20.097 J/mL)]; Tb is body temperature (°C); and Ta is chamber air temperature (°C). Only data from animals fitted with temperature-sensitive radio transmitters were used for these calculations.
| Season and sex | Thermal conductance | ||||
|---|---|---|---|---|---|
| Wet ± s.d. | (n) | Dry ± s.d. | (n) | ||
| Summer | |||||
| Captive, both sexes | 0.0256 ± 0.0028 | (18) | 0.0246 ± 0.0019 | (12) | |
| Winter | |||||
| Captive, female | 0.0172 ± 0.0023 | (10) | 0.0161 ± 0.0027 | (6) | |


[Pg 11] Cmw was calculated for each season from metabolic measurements made at all air temperatures below Tlc (Table 3). Because evaporative water loss was not measured at temperatures below freezing, Cmd was calculated only from metabolic determinations made at air temperatures between Tlc and 0°C. There was no difference between males and females in summer for either Cmw or Cmd (mL O2·g-1·h-1·°C-1). Data for each sex were combined to give a summer average of 0.0256 ± 0.0028 for Cmw, and 0.0246 ± 0.0019 for Cmd (Table 3). These summer conductances were 49% higher (p<0.005) than those calculated for winter females (0.0172 ± 0.0023, and 0.0161 ± 0.0027 for Cmw and Cmd, respectively; Table 3). Cmw and Cmd were not different from each other in either summer or winter, which indicated that in both seasons evaporative water loss contributed very little to heat dissipation at temperatures below Tn. Comparisons of thermal conductances calculated on the basis of metabolic body size (Mellen, 1963) gave the same results.
[Pg 11] Cmw was calculated for each season using metabolic measurements taken at all air temperatures below Tlc (Table 3). Since we didn't measure evaporative water loss at temperatures below freezing, Cmd was only calculated from metabolic data recorded at air temperatures between Tlc and 0°C. There was no difference between males and females in summer for either Cmw or Cmd (mL O2·g-1·h-1·°C-1). Data for both sexes were combined to provide a summer average of 0.0256 ± 0.0028 for Cmw, and 0.0246 ± 0.0019 for Cmd (Table 3). These summer conductances were 49% higher (p<0.005) than those calculated for winter females (0.0172 ± 0.0023 for Cmw and 0.0161 ± 0.0027 for Cmd; Table 3). Cmw and Cmd showed no differences from each other in either summer or winter, indicating that in both seasons, evaporative water loss played a minimal role in heat dissipation at temperatures below Tn. Comparisons of thermal conductances calculated based on metabolic body size (Mellen, 1963) yielded the same results.
Evaporative water loss increased as chamber temperature increased in both summer and winter (Figures 4, 5). In summer, the pattern of increase was different for females and males. Polynomial regressions for trapped and captive males produced equations that describe a concave relationship between Ta and evaporative water loss, whereas the equation for females describes a sigmoid curve (Table 4; Figure 4). For females, water loss increased rapidly at temperatures above 25°C (Figure 4). The intercepts and coefficients of the X, X2, and X3 terms of the polynomial regression equations (Table 4) were compared (t-tests) to determine if they differed from each other. The coefficients in the equation for trapped males differed from those for captive females in the X2 (p<0.05) and X3 (p<0.025) terms. The intercept and coefficients of the equation for captive males, however, were not different from those for either captive females or trapped males. Although this lack of difference is understandable in the case of trapped males, where the shape of the two curves is similar (concave), it is not so clear for the sigmoid curve of captive females (Figure 4). Perhaps the lack of difference in this case is simply due to the small number of observations available for captive males (n = 10; Table 4). Nonetheless, in summer at 35°C, both captive and trapped males relied less on evaporative cooling than did captive females (Figure 4).
Evaporative water loss increased as the chamber temperature rose in both summer and winter (Figures 4, 5). In summer, the pattern of increase was different for females and males. Polynomial regressions for trapped and captive males produced equations that show a concave relationship between Ta and evaporative water loss, while the equation for females indicates a sigmoid curve (Table 4; Figure 4). For females, water loss increased quickly at temperatures above 25°C (Figure 4). The intercepts and coefficients of the X, X2, and X3 terms of the polynomial regression equations (Table 4) were compared using t-tests to see if they were different from each other. The coefficients in the equation for trapped males differed from those for captive females in the X2 (p<0.05) and X3 (p<0.025) terms. However, the intercept and coefficients of the equation for captive males were not different from those for either captive females or trapped males. While this lack of difference makes sense for trapped males, where the shape of the two curves is similar (concave), it is less clear for the sigmoid curve of captive females (Figure 4). The lack of difference in this case may simply be due to the small number of observations available for captive males (n = 10; Table 4). Nonetheless, in summer at 35°C, both captive and trapped males relied less on evaporative cooling than captive females did (Figure 4).
In winter, males and females had similar rates of evaporative water loss across the full range of temperatures tested (Figure 5). Therefore, data for both sexes were combined. The intercept and coefficients of this equation (Table 4) did not differ from those for summer females, but they did differ from those in the regression for trapped males in the X2 (p<0.05) and X3 (p<0.025) terms. As was the case for females in summer, rates of water loss for winter animals increased most rapidly at temperatures above 25°C (Figure 5).
In winter, males and females experienced similar rates of evaporative water loss across all tested temperatures (Figure 5). Therefore, the data for both sexes were combined. The intercept and coefficients of this equation (Table 4) were the same as those for summer females, but they differed from the regression for trapped males in the X2 (p<0.05) and X3 (p<0.025) terms. Just like with summer females, the water loss rates for winter animals increased most quickly at temperatures above 25°C (Figure 5).

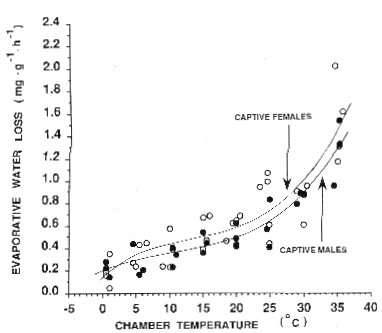
| Season and sex | Equation | (n) | R2 | |||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Summer | ||||||||||
| Trapped male | Y = | 0.1899 | + | 0.0114 X | + | 0.0011 X2 | - | 0.00002 X3 | (32) | 0.86 |
| SEE | 0.0885 | 0.0223 | 0.0015 | 0.00003 | ||||||
| Captive male | Y = | 0.2174 | + | 0.0192·X | + | 0.0009·X2 | - | 0.00003·X3 | (10) | 0.73 |
| SEE | 0.3983 | 0.0834 | 0.0048 | 0.00008 | ||||||
| Captive female | Y = | 0.0127 | + | 0.0943·X | - | 0.0060·X2 | + | 0.00013·X3 | (31) | 0.64 |
| SEE | 0.2218 | 0.0547 | 0.0036 | 0.00006 | ||||||
| Winter | ||||||||||
| Captive, both sexes | Y = | 0.1550 | + | 0.0426·X | - | 0.0025·X2 | + | 0.00006·X3 | (57) | 0.80 |
| SEE | 0.0734 | 0.0192 | 0.0013 | 0.00002 | ||||||
Body temperatures in Figure 6 are those recorded during metabolic measurements from animals equipped with surgically implanted, temperature-sensitive radio transmitters. Each point was recorded during the lowest level of oxygen consumption at each Ta. In both summer and winter, Tb's were lowest during metabolic measurements at Ta's around Tlc. At Ta's below Tlc, Tb's increased (Figure 6), which is an unusual [Pg 13] response. Under similar conditions, other procyonids either maintain a nearly constant Tb or allow it to fall slightly (Müller and Kulzer, 1977; Chevillard-Hugot et al., 1980; Müller and Rost, 1983; Chevalier, 1985). For our raccoons, confinement in the metabolism chamber at low temperatures must have stimulated a greater than necessary increase in metabolic rate such that heat production exceeded heat loss, which caused Tb to become elevated.
Body temperatures in Figure 6 were recorded during metabolic measurements from animals fitted with surgically implanted, temperature-sensitive radio transmitters. Each data point was captured during the lowest level of oxygen consumption at each Ta. In both summer and winter, Tb was lowest during metabolic measurements at Ta around Tlc. At Ta below Tlc, Tb increased (Figure 6), which is an unusual response. Under similar conditions, other procyonids either maintain nearly constant Tb or allow it to drop slightly (Müller and Kulzer, 1977; Chevillard-Hugot et al., 1980; Müller and Rost, 1983; Chevalier, 1985). For our raccoons, being confined in the metabolism chamber at low temperatures must have triggered an unnecessary increase in metabolic rate, resulting in heat production exceeding heat loss, which caused Tb to rise.

| Season and sex | Equation | (n) | R2 | SEE | I | |
|---|---|---|---|---|---|---|
| a | b | |||||
| Summer | ||||||
| Trapped male | Y = 1.09 - 0.0281·X | (30) | 0.64 | 0.0353 | 0.0040 | 38.8 |
| Captive male | Y = 0.97 - 0.0258·X | (12) | 0.91 | 0.0235 | 0.0025 | 37.6 |
| Captive female | Y = 1.04 - 0.0251·X | (29) | 0.78 | 0.0288 | 0.0026 | 41.1 |
| Winter | ||||||
| Captive, both sexes | Y = 0.68 - 0.0193·X | (36) | 0.68 | 0.0157 | 0.0023 | 35.2 |
During summer, Tlc for male raccoons was 20°C, whereas for females it was 25°C (Figure 2). Regression equations calculated to describe oxygen consumption at Ta's below Tlc are presented in Table 5. For three groups of summer animals, slopes of regressions are identical. This indicates that minimum conductances of these three groups were equivalent. Intercepts of these equations are different, which suggests a difference in metabolic cost of thermoregulation between these groups (Figure 2); captive males had a lower intercept than either trapped males (p<0.005) or captive females (p<0.05), but there was no difference in intercepts of captive females and trapped males. These regression equations, therefore, also were derived using values of oxygen consumption expressed in terms of metabolic body mass (Mellen, 1963). Relationships between intercepts of these equations are different than those for regressions in Table 5. Intercept for females was intermediate to, and not different from, those of the two groups of males. However, captive males still had a lower intercept than trapped males (p<0.025). Thus, in summer, thermoregulatory metabolism was less expensive for captive than for trapped males, and in spite of a 5°C difference in their Tlc's (Figure 2), captive males and females had similar thermoregulatory costs.
During summer, the lower critical temperature (Tlc) for male raccoons was 20°C, while for females it was 25°C (Figure 2). The regression equations that describe oxygen consumption at ambient temperatures (Ta) below Tlc are shown in Table 5. For the three groups of summer animals, the slopes of the regressions are the same. This means that the minimum conductances for these three groups were equivalent. The intercepts of these equations differ, indicating a variation in the metabolic cost of thermoregulation among these groups (Figure 2); captive males had a lower intercept than both trapped males (p<0.005) and captive females (p<0.05), but there was no difference in the intercepts between captive females and trapped males. Therefore, these regression equations were also based on oxygen consumption values expressed as metabolic body mass (Mellen, 1963). The relationships between the intercepts of these equations are different from those for the regressions in Table 5. The intercept for females was in between and not significantly different from those of the two groups of males. However, captive males still had a lower intercept than trapped males (p<0.025). Thus, in summer, the thermoregulatory metabolism was less costly for captive than for trapped males, and despite a 5°C difference in their Tlc's (Figure 2), captive males and females shared similar thermoregulatory costs.
Regression lines for three groups of animals in summer extrapolate to zero metabolism at values equivalent to, or greater than, normal Tb; 38.8°C for trapped males, 37.6°C for captive males, and 41.1°C for captive females (Table 5). Thus, all three groups had minimized thermal conductance at Ta's below Tlc (Scholander et al., 1950b; McNab, 1980b). Minimum wet thermal conductance calculated for raccoons in summer with Eq. 4 (Table 3) is numerically similar to these "slope" values (Table 5), and it was, therefore, considered to be the best estimate of Cmw for Procyon lotor during that season (0.0256 mL O2·g-1·h-1·°C-1).
Regression lines for three groups of animals in summer show that metabolism reaches zero at temperatures equal to or higher than their normal body temperature; 38.8°C for trapped males, 37.6°C for captive males, and 41.1°C for captive females (Table 5). Thus, all three groups had reduced thermal conductance at air temperatures below Tlc (Scholander et al., 1950b; McNab, 1980b). The minimum wet thermal conductance calculated for raccoons in summer with Eq. 4 (Table 3) is numerically similar to these "slope" values (Table 5), and is therefore regarded as the best estimate of Cmw for Procyon lotor during that season (0.0256 mL O2·g-1·h-1·°C-1).
During winter Tlc for both sexes decreased to 11°C (Figure 3). Regression equations of thermoregulatory metabolism for males and females in winter are not different from each other in either slope or intercept. These data, therefore, were combined into a single equation (Table 5). Slope and intercept of this equation are both lower (p<0.005 and p<0.05, respectively) than those for summer animals (Table 5). Identical results were obtained from comparisons using regressions derived from oxygen consumption expressed in terms of metabolic body mass (Mellen, 1963). Thermoregulatory costs at any temperature below 20°C were lower for winter than summer animals (Figures 2, 3).
During winter, Tlc for both males and females dropped to 11°C (Figure 3). The regression equations for thermoregulatory metabolism in males and females during winter are similar in both slope and intercept. Therefore, these data were combined into a single equation (Table 5). The slope and intercept of this equation are both lower (p<0.005 and p<0.05, respectively) compared to summer animals (Table 5). Identical results were found when comparing regressions based on oxygen consumption relative to metabolic body mass (Mellen, 1963). Thermoregulatory costs at any temperature below 20°C were lower for winter animals than for summer animals (Figures 2, 3).
| Group | Equation | (n) | R2 | I |
|---|---|---|---|---|
| A | Y = 0.63 - 0.0158·X | (10) | 0.66 | 40.1 |
| B | Y = 0.72 - 0.0226·X | (11) | 0.71 | 32.1 |
| C | Y = 0.69 - 0.0200·X | (15) | 0.79 | 34.7 |

The regression line for Procyon lotor in winter (Table 5) extrapolates to zero metabolism at 35.2°C, which is below normal Tb (Figures 6, 7). This suggests that not all raccoons measured in winter minimized thermoregulatory metabolism or conductances at Ta's below Tlc (Scholander et al., 1950b; McNab, 1980b). To assess this possibility, data for these animals were divided into three groups: (A) females with radio transmitters, (B) females without radio transmitters, and (C) males (Table 6). Regression equations of metabolism below Tlc were derived for each group, and based on extrapolated Tb's at zero metabolism, only the two females with implanted radio transmitters (group A) minimized thermoregulatory metabolism and conductance. Had animals in groups B and C also minimized their thermal conductances, while retaining their measured metabolic rates, their rates of heat production would have been disproportionately higher than their rates of heat loss. Equation 4 predicts that under these conditions their body temperatures would have been elevated to 42.0°C and 40.4°C, respectively. Thus, in order to avoid such a large increase in body temperature, animals in groups B and C increased their thermal conductances in preference to lowering their metabolic [Pg 16] rates. The regression equation of thermoregulatory metabolism for all winter animals (Table 5), therefore, overestimates minimum metabolic cost of temperature regulation below Tlc, and its slope underestimates Cmw. Consequently, the best estimate of Cmw for Procyon lotor in winter is the value calculated for group A animals with Eq. 4 (0.0172 mL O2·g-1·h-1·°C-1; Table 3), and the minimum cost of thermoregulatory metabolism at any Ta below Tlc is best estimated by substituting this value into Eq. 4 and solving for Ḣr.
The regression line for Procyon lotor in winter (Table 5) shows that metabolism reaches zero at 35.2°C, which is below the normal body temperature (Tb) (Figures 6, 7). This means that not all raccoons measured in winter reduced their thermoregulatory metabolism or conductances at ambient temperatures (Ta) below critical temperature (Tlc) (Scholander et al., 1950b; McNab, 1980b). To investigate this further, the data were split into three groups: (A) females with radio transmitters, (B) females without radio transmitters, and (C) males (Table 6). Regression equations for metabolism below Tlc were calculated for each group. Based on the projected Tb at zero metabolism, only the two females with implanted radio transmitters (group A) managed to minimize their thermoregulatory metabolism and conductance. If the animals in groups B and C had also minimized their thermal conductances while keeping their measured metabolic rates, their heat production would have been much higher than their heat loss. Equation 4 indicates that under these conditions, their body temperatures would have risen to 42.0°C and 40.4°C, respectively. Therefore, to avoid such a significant increase in body temperature, the animals in groups B and C raised their thermal conductances instead of lowering their metabolic rates. The regression equation for thermoregulatory metabolism for all winter animals (Table 5) thus overestimates the minimum metabolic cost of temperature regulation below Tlc and its slope underestimates Cmw. As a result, the most accurate estimate of Cmw for Procyon lotor in winter is the value calculated for group A animals with Eq. 4 (0.0172 mL O2·g-1·h-1·°C-1; Table 3), and the minimum cost of thermoregulatory metabolism at any Ta below Tlc is best estimated by substituting this value into Eq. 4 and solving for Ḣr.
In both summer and winter, Tb's increased during metabolic measurements at Ta's above Tlc (Figure 6). This response also was seen during metabolic measurements conducted on other procyonids (Müller and Kulzer, 1977; Chevillard-Hugot et al., 1980; Müller and Rost, 1983; Chevalier, 1985).
In both summer and winter, Tb increased during metabolic measurements at Ta levels above Tlc (Figure 6). This reaction was also observed during metabolic measurements done on other procyonids (Müller and Kulzer, 1977; Chevillard-Hugot et al., 1980; Müller and Rost, 1983; Chevalier, 1985).
During summer our data suggested that the upper critical temperature (Tuc) was higher than 35°C. The lowest rates of oxygen consumption at Ta = 35°C occurred after 1.5 to 2.5 hours of exposure to that temperature. Prolonged exposure to this temperature in summer did not make animals restless, and their rate of oxygen consumption was very stable throughout each measurement. Body temperature responses at Ta = 35°C were recorded from two males and two females that had implanted radio transmitters. With the exception of one male, Tb's were maintained near 38°C (Figure 6). The one exception (a male) maintained its Tb at 39.3°C. At Ta = 35°C, summer males had rates of evaporative water loss that were lower than those of summer females (Figure 4). At this temperature, males dissipated 35% ± 6% and females 56% ± 18% of their metabolic heat via evaporative water loss. Thus, at Ta = 35°C, males must have utilized modes of heat transfer other than evaporative cooling (convective and conductive heat transfer) to a greater extent than females.
During the summer, our data indicated that the upper critical temperature (Tuc) was above 35°C. The lowest levels of oxygen consumption at Ta = 35°C were observed after 1.5 to 2.5 hours of exposure to that temperature. Prolonged exposure to this temperature in summer didn't cause the animals to be restless, and their oxygen consumption rates remained very stable throughout each measurement. Body temperature responses at Ta = 35°C were recorded from two males and two females that had implanted radio transmitters. With the exception of one male, Tb's were maintained around 38°C (Figure 6). The one exception (a male) maintained its Tb at 39.3°C. At Ta = 35°C, summer males had lower rates of evaporative water loss than summer females (Figure 4). At this temperature, males lost 35% ± 6% and females 56% ± 18% of their metabolic heat through evaporative water loss. Therefore, at Ta = 35°C, males must have relied on other forms of heat transfer besides evaporative cooling (like convective and conductive heat transfer) more than females did.
Body temperature, evaporative water loss, and metabolic data indicated that, in winter, Tuc was very close to 35°C. In winter, the lowest level of oxygen consumption was recorded during the first hour after the chamber had reached Ta = 35°C. Unlike summer, animals became restless after the first hour at 35°C, at which point their oxygen consumption increased and showed a high degree of variability. Body temperature responses at 35°C were recorded from both females that had implanted radio transmitters. In one case, Tb rose from 37.9°C at the end of the first hour to 40.5°C by the end of the second hour, and as it did not show signs of leveling off, we terminated the experiment. We exposed that same animal to Ta = 35°C one other time during winter. In that instance, its Tb rose to 40.0°C during the first 30 minutes and was maintained at that level for three hours with no apparent distress. The other female elevated its Tb from 37.3°C to 39.0°C during the second hour at Ta = 35°C and maintained its Tb at that level for two hours. Thus, during winter, prolonged exposure to Ta = 35°C stimulated more of an increase in Tb than it did in summer. During winter, both males and females increased evaporative water loss at Ta = 35°C (Figure 5) but only to the extent that they dissipated 35% ± 10% of their metabolic heat production. Thus, even in winter, convective and conductive heat transfers were still the most important modes of heat loss at this temperature.
Body temperature, evaporative water loss, and metabolic data showed that in winter, Tuc was very close to 35°C. During winter, the lowest oxygen consumption was recorded in the first hour after the chamber reached Ta = 35°C. Unlike in summer, the animals became restless after the first hour at 35°C, at which point their oxygen consumption increased and became highly variable. Body temperature responses at 35°C were recorded from both females with implanted radio transmitters. In one case, Tb rose from 37.9°C at the end of the first hour to 40.5°C by the end of the second hour, and since it didn’t level off, we ended the experiment. We exposed that same animal to Ta = 35°C again in winter. During that exposure, its Tb rose to 40.0°C within the first 30 minutes and was sustained at that level for three hours with no signs of distress. The other female increased its Tb from 37.3°C to 39.0°C during the second hour at Ta = 35°C and kept its Tb at that level for two hours. Therefore, in winter, prolonged exposure to Ta = 35°C triggered a greater rise in Tb than in summer. During winter, both males and females increased evaporative water loss at Ta = 35°C (Figure 5) but only managed to dissipate 35% ± 10% of their metabolic heat production. Hence, even in winter, convective and conductive heat transfers remained the primary methods of heat loss at this temperature.
The daily cycle of raccoon Tb's during summer and winter are presented in Figure 7. In general, Tb's showed a marked circadian cycle in phase with photoperiod. Tb's rose above 38°C for several hours each night but remained below 38°C during daytime. During summer, with the exception of one female whose record was not typical (Figure 7), Tb's rose above 38°C shortly after sunset, whereas in winter Tb's did not rise above 38°C until several hours after sunset. Once Tb was elevated it usually remained so until just before or after sunrise (Figure 7). During summer, Tb was above 38°C for 85% or more of the time between sunset and sunrise (87% for the female with the typical body temperature pattern, and 85% and 98% for males), whereas in winter it was elevated for only 47%-78% of the time between sunset and sunrise (47% and 61% for females, and 67% and 78% for males). During night, Tb would oscillate between 38°C and about 39°C, such that two peak values occurred. These peak values presumably corresponded to two periods of heightened nighttime activity. During summer, one of these peaks occurred before and the other after 24:00 hours, whereas in winter both peaks occurred after 24:00 hours. With the exception of one female in winter (Figure 7), the lowest Tb of the day for both sexes was near 37°C, and this typically occurred during daytime (Figure 7).
The daily patterns of raccoon Tb's during summer and winter are detailed in Figure 7. Generally, Tb's displayed a clear circadian rhythm aligned with the light-dark cycle. Tb's rose above 38°C for several hours each night but stayed below 38°C during the day. In the summer, except for one female whose data was unusual (Figure 7), Tb's climbed above 38°C shortly after sunset, while in winter, Tb's didn’t exceed 38°C until a few hours after sunset. Once Tb increased, it typically stayed elevated until right before or after sunrise (Figure 7). During summer, Tb was above 38°C for 85% or more of the time between sunset and sunrise (87% for the female with the typical body temperature pattern, and 85% and 98% for males), while in winter it was increased for only 47%-78% of that time (47% and 61% for females, and 67% and 78% for males). At night, Tb fluctuated between 38°C and around 39°C, resulting in two peak readings. These peaks likely related to two times of increased nighttime activity. In summer, one peak occurred before midnight and the other after, whereas in winter, both peaks happened post-midnight. Except for one female in winter (Figure 7), the lowest Tb for both sexes was around 37°C, usually occurring during the day (Figure 7).
Basal metabolism represents the minimum energy required by a mammal to maintain endothermy and basic homeostasis (Lusk, 1917:141; Kleiber, 1932, 1961:251; Benedict, 1938:191-215; Brody, 1945:59; Robbins, 1983:105-111). Mammals with lower than predicted Ḣb maintain endothermy and enjoy its attendant advantages at a discount, whereas others, with rates that are higher than predicted, pay a premium [Pg 17] (Calder, 1987). Such variation in Ḣb appears to be tied to ecological circumstances rather than taxonomic affinities (Vogel, 1980; McNab, 1986a, 1988a, 1989), and depending on environmental conditions, each rate provides an individual with various advantages and limitations. During the course of evolution, therefore, each species' Ḣb evolves to provide it with the best match between its energy requirements for continuous endothermy, its food supply, and the thermal characteristics of its environment.
Basal metabolism refers to the minimum energy that a mammal needs to maintain its body heat and basic stability (Lusk, 1917:141; Kleiber, 1932, 1961:251; Benedict, 1938:191-215; Brody, 1945:59; Robbins, 1983:105-111). Mammals with a lower than expected Ḣb can maintain their body heat and enjoy the benefits that come with it at a lower cost, while those with higher than expected rates have to pay more (Calder, 1987). This variation in Ḣb seems to be related to ecological factors instead of taxonomical relationships (Vogel, 1980; McNab, 1986a, 1988a, 1989). Depending on environmental conditions, each rate offers individuals different advantages and disadvantages. Over time, each species' Ḣb evolves to give it the best balance between its energy needs for sustained body heat, its food availability, and the temperature features of its surroundings.
Male raccoons trapped in summer had higher Ḣb's than our captive animals in any season (Table 2). The higher rate of metabolism of these trapped males could have been due to the stress of captivity or to the fact that "wild" animals actually may have higher metabolic rates than those that have adjusted to captivity. If the latter is true, then our data for captive animals underestimated the actual energy cost of maintenance metabolism for Procyon lotor in the wild. At present, we have no way of determining which of these alternatives is true.
Male raccoons trapped in summer had higher Ḣb's than our captive animals in any season (Table 2). The increased metabolism in these trapped males might have been due to the stress of captivity or the fact that "wild" animals could have higher metabolic rates than those that have acclimated to captivity. If that’s the case, then our data for captive animals could underestimate the actual energy costs of maintenance metabolism for Procyon lotor in the wild. Right now, we have no way to determine which of these possibilities is accurate.
In some temperate-zone mammals, Ḣb is elevated in winter, which presumably increases their "cold-hardiness." Conversely, lower summer metabolism is considered to be a mechanism that reduces the potential for heat stress. Such seasonal variation in Ḣb has been found in several species: collard peccary, Tayassu tajacu (Zervanos, 1975); antelope jackrabbit, Lepus alleni (Hinds, 1977); desert cottontail, Sylvilagus audubonii (Hinds, 1973); and, perhaps, cold-acclimatized rat, Rattus norvegicus (Hart and Heroux, 1963). Unlike these species, our captive raccoons showed no seasonal variation in Ḣb (Table 2). Instead, raccoons achieved "cold-hardiness" in winter and reduced their potential for heat stress in summer with a large seasonal change in thermal conductance (Table 3).
In some temperate mammals, Ḣb increases in winter, which likely boosts their ability to handle cold. On the other hand, a lower metabolism in summer is thought to help reduce the risk of heat stress. This seasonal change in Ḣb has been observed in several species: collard peccary, Tayassu tajacu (Zervanos, 1975); antelope jackrabbit, Lepus alleni (Hinds, 1977); desert cottontail, Sylvilagus audubonii (Hinds, 1973); and possibly in cold-acclimatized rat, Rattus norvegicus (Hart and Heroux, 1963). Unlike these animals, our captive raccoons did not show any seasonal changes in Ḣb (Table 2). Instead, raccoons became "cold-hardy" in winter and minimized their heat stress potential in summer by significantly changing their thermal conductance (Table 3).
| Species | Body Mass (g) | Basal[a] metabolism | Minimum[b] conductance | Tb[c] | Tn[d] | References | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Meas | Hbr | Meas | Cmwr | α | ρ | Tlc | Tuc | ||||||||
| Bassariscus astutus | 865 | 0.43 | 0.68 | 0.0288[e] | 0.85 | 37.6 | 23 | 35.5 | Chevalier (1985) | ||||||
| Procyon cancrivorus | 1160 | 0.40 | 0.69 | 0.0368[e] | 1.25 | 26 | Scholander et al. (1950b, c) | ||||||||
| Potos flavus | 2030 | 0.36 | 0.51 | McNab (1978a) | |||||||||||
| Potos flavus | 2400 | 0.32 | 0.65 | 38.1 | 36.0 | 23 | 30 | Müller and Kulzer (1977) | |||||||
| Potos flavus | 2600 | 0.34 | 0.71 | 0.0200[f] | 1.02 | 23 | 33 | Müller and Rost (1983) | |||||||
| Nasua nasua | 3850 | 0.26 | 0.60 | 0.0200[f] | 1.24 | 38.3 | 36.4 | 25 | 33 | Chevillard-Hugot et al. (1980) | |||||
| Nasua nasua | 4847 | 0.33 | 0.79 | 0.0238[e] | 1.65 | 39.1 | 37.9 | 30 | 35 | Mugaas et al. (in prep.) | |||||
| Nasua narica | 5554 | 0.25 | 0.62 | 0.0208[e] | 1.55 | 38.9 | 37.4 | 25 | 35 | ||||||
| Nasua narica | 4150 | 0.42 | 1.20 | 0.0341[e] | 2.20 | Scholander et al. (1950b, c) | |||||||||
| 0.0224[g] | 1.45 | ||||||||||||||
| Procyon lotor | This study | ||||||||||||||
| Summer | |||||||||||||||
| Trapped male | 4400 | 0.54 | 1.28 | 20 | |||||||||||
| Captive male | 4790 | 0.46 | 1.07 | 0.0256[f] | 1.77 | 38.4 | 37.5 | 20 | |||||||
| Captive female | 4670 | 0.42 | 1.02 | 0.0256[f] | 1.79 | 38.2 | 37.6 | 25 | |||||||
| Winter | |||||||||||||||
| Captive male | 5340 | 0.47 | 1.17 | 38.6 | 38.6 | 11 | |||||||||
| Captive female | 4490 | 0.46 | 1.10 | 0.0172[f] | 1.15 | 38.3 | 37.3 | 11 | |||||||
[b] Meas is measured minimum thermal conductance (mL O2/g/h/°C). Cmwr is the ratio of measured to predicted minimum thermal conductance where the predicted value is calculated from Cm = 1.0·m-0.5 (McNab and Morrison, 1963; Herreid and Kessel, 1967), and m is body mass in grams.
[c] Tb is body temperature during the active (α) and rest (ρ) phases of the daily cycle (°C).
[d] Tn is the thermoneutral zone as defined by the lower (Tlc) and upper (Tuc) critical temperatures (°C).
[e] Conductance calculated as the slope of the line describing oxygen consumption at temperatures below the lower critical temperature.
[f] Conductance calculated from Cmw = Ḣr/(Tb - Ta), where Ḣr is resting metabolic rate at temperatures below Tlc, and other symbols are as described elsewhere.
[g] Inactive-phase thermal conductance: estimated from Scholander et al. (1950b), assuming that active-phase thermal conductance is 52% higher than values determined during the inactive phase (Aschoff, 1981).
Procyon lotor has a much higher mass-specific Ḣb than other procyonids (Table 7). To quantify the magnitude of this difference, we compared the measured value for Procyon lotor [Pg 18] with one calculated for it from a mass-specific least-squares regression equation (Eq. 6; R2 = 0.78) derived from data for those procyonids with lower than predicted Ḣb: Potos flavus, Procyon cancrivorus, Nasua nasua, Nasua narica, and Bassariscus astutus (Table 7).
Procyon lotor has a significantly higher mass-specific Ḣb than other procyonids (Table 7). To measure this difference, we compared the actual value for Procyon lotor [Pg 18] with a value calculated from a mass-specific least-squares regression equation (Eq. 6; R2 = 0.78) based on data from procyonids that had lower than expected Ḣb: Potos flavus, Procyon cancrivorus, Nasua nasua, Nasua narica, and Bassariscus astutus (Table 7).
| Ḣb = 2.39·m-0.25 | Eq. 6 |
Ḣb in Eq. 6 is basal metabolism (mL O2·g-1·h-1) and m is body mass (g). Measured values of Ḣb for Procyon lotor were 1.45 to 1.86 times greater than those predicted for it by Eq. 6 (Table 8).
Ḣb in Eq. 6 is the basal metabolism (mL O2/g·h) and m is body mass (g). The measured values of Ḣb for Procyon lotor were 1.45 to 1.86 times higher than what Eq. 6 predicted for it (Table 8).
| Season and sex | Predicted | Measured/Predicted | |
|---|---|---|---|
| Summer | |||
| Trapped male | 0.29 | 1.86 | |
| Captive male | 0.29 | 1.59 | |
| Captive female | 0.29 | 1.45 | |
| Winter | |||
| Captive male | 0.28 | 1.68 | |
| Captive female | 0.29 | 1.59 | |
Background.—With respect to Ḣb, McNab (1986a:1) maintains that "the influence of climate is confounded with the influence of food habits," and that departures from the Kleiber (1961) "norm" are best correlated with diet. Although this does appear to be the case for diet specialists, the analysis is not so clear-cut for omnivorous species (McNab, 1986a). His analysis also indicates that an animal's "behavior" (i.e., whether it is terrestrial, arboreal, subterranean, aquatic, etc.), secondarily modifies the influence of food habits on Ḣb. For example, terrestrial frugivores have Ḣb's that are very near predicted values, whereas arboreal frugivores have rates that are much lower than predicted (McNab, 1986a).
Background.—Regarding Ḣb, McNab (1986a:1) argues that "the impact of climate is mixed up with the impact of eating habits," and that deviations from the Kleiber (1961) "norm" are most strongly linked to diet. While this seems true for specialists in diet, the situation is less clear for omnivorous species (McNab, 1986a). His analysis also shows that an animal's "behavior" (like whether it lives on land, in trees, underground, in water, etc.) also influences how food habits affect Ḣb. For instance, terrestrial frugivores have Ḣb values that closely match predicted values, while arboreal frugivores show much lower rates than expected (McNab, 1986a).
| + | <20% by volume when found. | † | 1%-19% frequency of occurrence. |
| ++ | >20% by volume when found. | †† | 20%-50% frequency of occurrence. |
| ††† | >50% frequency of occurrence. | ||
| Food | Potos flavus | Procyon cancrivorus | Nasua nasua | Nasua narica | Bassariscus | Procyon lotor | ||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Mammalia | + | † | # | ++ | ††† | ++ | †† | |||||
| Aves | ++ | † | + | †† | ||||||||
| Birds' eggs | ††† | |||||||||||
| Reptilia | + | † | + | ††† | # | + | † | + | † | |||
| Amphibia | + | † | + | † | ||||||||
| Pices | ++ | †† | ++ | †† | ||||||||
| Insecta | ++ | † | + | ††† | ++ | ††† | # | + | †† | ++ | †† | |
| Arachnida | ++ | ††† | # | + | † | + | † | |||||
| Chilopoda | ++ | ††† | ||||||||||
| Diplopoda | # | + | † | |||||||||
| Crustacea | ++ | ††† | # | ++ | ††† | |||||||
| Mollusca | + | †† | # | + | †† | |||||||
| Annelida | # | + | † | |||||||||
| Nuts | ++ | †† | ||||||||||
| Grains | ++ | †† | ||||||||||
| Buds | + | † | ||||||||||
| Fruit | ++ | ††† | ++ | # | †† | ++ | ††† | |||||
| Leaves | + | † | ||||||||||
| Grass | + | † | ||||||||||
Food Habits of Procyonids.—Food habits of six procyonids for which metabolic data are available are presented in Table 9. All six species clearly have mixed diets. Compared to other species, Procyon lotor is highly catholic in its diet, taking food from almost twice as many categories as Nasua narica, three times as many as Procyon cancrivorus, Nasua nasua, and Bassariscus astutus, and nine times as many as Potos flavus.
Procyonid Eating Habits.—The eating habits of six procyonids for which metabolic data are available are presented in Table 9. All six species clearly have mixed diets. Compared to other species, Procyon lotor has a very varied diet, consuming food from almost twice as many categories as Nasua narica, three times as many as Procyon cancrivorus, Nasua nasua, and Bassariscus astutus, and nine times as many as Potos flavus.
[Pg 19] For those species for which food habit data are quantified, we used Eisenberg's (1981:247-251) substrate/feeding matrix method, where "substrate" is analogous to McNab's (1986a) "behavior," to construct the following feeding categories that are based on the major food groups utilized by each species (Table 9).
[Pg 19] For the species with quantified food habit data, we applied Eisenberg's (1981:247-251) substrate/feeding matrix method, where "substrate" is similar to McNab's (1986a) "behavior." This helped us create the following feeding categories based on the main food groups used by each species (Table 9).
1. Potos flavus: (1) arboreal/frugivore, insectivore.
2. Procyon cancrivorus: (1) semiaquatic/crustacivore, molluscivore, insectivore, piscivore, carnivore.
3. Nasua nasua: (1) terrestrial/insectivore, arachnidivore, carnivore, frugivore.
4. Bassariscus astutus: (1) terrestrial/carnivore, insectivore, frugivore.
5. Procyon lotor: (1) terrestrial/carnivore, granivore, frugivore, insectivore; and (2) semiaquatic/crustacivore, molluscivore, insectivore, piscivore, carnivore.
1. Potos flavus: (1) tree-dwelling/fruit eater, insect eater.
2. Procyon cancrivorus: (1) semi-aquatic/crustacean eater, mollusk eater, insect eater, fish eater, carnivore.
3. Nasua nasua: (1) ground-dwelling/insect eater, spider eater, carnivore, fruit eater.
4. Bassariscus astutus: (1) ground-dwelling/carnivore, insect eater, fruit eater.
5. Procyon lotor: (1) ground-dwelling/carnivore, seed eater, fruit eater, insect eater; and (2) semi-aquatic/crustacean eater, mollusk eater, insect eater, fish eater, carnivore.
Food Habits and Basal Metabolism.—The most important foods in the diet of Procyon lotor are vertebrates, nuts, seeds, and fruits (Table 9). These are the same foods that are eaten by those dietary specialists that have Ḣb's equivalent to, or higher than, values predicted for them by the Kleiber equation (McNab, 1986a). The most important foods in the diets of Potos flavus, Procyon cancrivorus, and Nasua nasua are invertebrates and fruit (Table 9), and these foods are eaten by dietary specialists that have lower than predicted Ḣb's (McNab, 1986a). Major foods in the diet of Bassariscus astutus are terrestrial vertebrates, insects, and fruit (Table 9). Dietary specialists that eat terrestrial vertebrates have higher than predicted Ḣb's, whereas those that feed on insects have Ḣb's that are lower than predicted (McNab, 1986a). Year-round utilization of vertebrates by Bassariscus astutus suggests that it also should have a metabolic rate that is equivalent to or higher than predicted, rather than lower (McNab, 1986a). However, perhaps year-round inclusion of insects in its diet (Martin et al., 1951; Taylor, 1954; Wood, 1954; Toweill and Teer, 1977; Trapp, 1978), plus water-and energy-conserving advantages of a low metabolic rate, each exert a stronger selective influence on Ḣb than do vertebrates in its diet.
Eating Habits and Metabolism.—The main foods in the diet of Procyon lotor are vertebrates, nuts, seeds, and fruits (Table 9). These are the same foods eaten by dietary specialists that have Ḣb values equal to or higher than those predicted for them by the Kleiber equation (McNab, 1986a). The key foods in the diets of Potos flavus, Procyon cancrivorus, and Nasua nasua are invertebrates and fruit (Table 9), and these foods are consumed by dietary specialists that have Ḣb values lower than predicted (McNab, 1986a). Major foods in the diet of Bassariscus astutus include terrestrial vertebrates, insects, and fruit (Table 9). Dietary specialists that eat terrestrial vertebrates typically have Ḣb values higher than predicted, while those that consume insects tend to have Ḣb values lower than expected (McNab, 1986a). The year-round consumption of vertebrates by Bassariscus astutus indicates that it should also have a metabolic rate that is equal to or higher than predicted, rather than lower (McNab, 1986a). However, the continuous inclusion of insects in its diet (Martin et al., 1951; Taylor, 1954; Wood, 1954; Toweill and Teer, 1977; Trapp, 1978), along with the water-and energy-conserving benefits of a low metabolic rate, might have a greater selective impact on Ḣb than the presence of vertebrates in its diet.
Summary.—The basal metabolic rate of these procyonids does appear to be influenced by diet. But, it is apparent from this family's evolutionary history and tropical origins that climate also has had a profound influence on its member's metabolism. The history of the family and the data presented here (Table 7) suggest that lower than predicted Ḣb is a feature that evolved very early as the primary metabolic adjustment to a tropical climate. From this perspective, it could be argued that climate would have been the major selective force determining Ḣb, whereas food habits would have had a secondary influence.
Summary.—The basal metabolic rate of these raccoons seems to be affected by their diet. However, it's clear from this family's evolutionary history and tropical origins that climate has also played a significant role in shaping their metabolism. The family's history and the data presented here (Table 7) suggest that the lower than expected Ḣb is a characteristic that developed very early as the main metabolic adaptation to a tropical environment. From this viewpoint, one could argue that climate was the primary selective factor influencing Ḣb, while dietary habits had a secondary impact.
Background.—McNab (1980a) suggested that if food is not restricted during an animal's reproductive period, the factor that will limit growth and reproduction will be the rate at which energy can be used in growth and development. Under these conditions, an increase in Ḣb would actually increase rmax because it would provide a higher rate of biosynthesis, a faster growth rate, and a shorter generation time. Hennemann (1983) tested McNab's (1980a) premise and found a significant correlation between rmax and metabolic rate, independent of body size, for 44 mammal species. A low correlation coefficient for this relationship, however, indicated to him (Hennemann, 1983) that factors such as (1) food supply, (2) thermal characteristics of the environment, and (3) brain size also contribute toward shaping a species' reproductive potential, particularly when these factors strongly influence rates of biosynthesis or growth or for some reason alter generation time. Results of our estimates of rmax for procyonids are presented in Table 10.
Background.—McNab (1980a) suggested that if food isn’t restricted during an animal's reproductive period, the limiting factor for growth and reproduction would be how quickly energy can be utilized for growth and development. In this scenario, an increase in Ḣb would actually raise rmax because it would allow for a higher rate of biosynthesis, faster growth, and shorter generation times. Hennemann (1983) tested McNab's premise and found a significant correlation between rmax and metabolic rate, regardless of body size, among 44 mammal species. However, a low correlation coefficient for this relationship led him (Hennemann, 1983) to conclude that factors like (1) food supply, (2) thermal characteristics of the environment, and (3) brain size also play a role in determining a species' reproductive potential, especially when these factors significantly impact biosynthesis or growth rates, or somehow affect generation time. Our estimates of rmax for procyonids are presented in Table 10.
Procyon lotor.—This species had the highest Ḣb and Dd, and also had the highest rmax (1.34; Table 10). Such a high rmax may infer that this trait evolved under conditions where food and temperature were not limiting to reproduction. Under these conditions selection could have favored those reproductive characteristics sensitive to a higher Ḣb (biosynthesis, growth, and generation time; McNab, 1980a). Procyon lotor's high reproductive potential is due to its early age of first female reproduction and its large litter size, characteristics that may reflect metabolically driven increases in both biosynthesis and growth.
Procyon lotor.—This species had the highest Ḣb and Dd, as well as the highest rmax (1.34; Table 10). Such a high rmax suggests that this trait developed when food and temperature were not limiting factors for reproduction. In these conditions, natural selection might have favored reproductive traits that are sensitive to a higher Ḣb (biosynthesis, growth, and generation time; McNab, 1980a). Procyon lotor's strong reproductive potential comes from its early age of first female reproduction and its large litter size, features that likely reflect increases in biosynthesis and growth driven by metabolic factors.
Bassariscus astutus.—This species has a low Ḣb but an rmax that was 124% of expected (Table 10). This suggests that rmax evolved under conditions where food and temperature were not limiting to reproduction. Reduced litter size should restrict this species' reproductive potential and may be a reflection of its low Ḣb. The factor that is responsible for increasing its reproductive potential, however, is its early age of first female reproduction. Bassariscus astutus is the smallest of these procyonids, and even though it has a low Ḣb, its small mass may contribute to its ability to reach adult size and sexual maturity in its first year. The high quality of its diet (a high proportion of small vertebrates; Table 9) also may be a factor that is permissive to early female reproduction. Thus, small body size and diet may be factors that have allowed this species to evolve a higher than expected reproductive potential in spite of its low Ḣb.
Bassariscus astutus.—This species has a low Ḣb but an rmax that was 124% of what was expected (Table 10). This indicates that rmax evolved in conditions where food and temperature didn't limit reproduction. The smaller litter size should limit this species' reproductive potential and may reflect its low Ḣb. However, what boosts its reproductive potential is the early age of first female reproduction. Bassariscus astutus is the smallest of these raccoon relatives, and even with its low Ḣb, its small size may help it reach adult size and sexual maturity within its first year. The high quality of its diet (a high proportion of small vertebrates; Table 9) might also contribute to early female reproduction. Therefore, its smaller body size and diet may be factors that have allowed this species to develop a reproductive potential that's higher than expected despite its low Ḣb.
Nasua narica.—This species is one of the largest procyonids (Table 7), and it possesses characteristics that should limit its reproductive potential: lower than predicted Ḣb (Table 7), a relatively low-quality diet (Kaufmann, 1962:182-198; Table 9), and delayed time of first reproduction (Table 10). In spite of this, Nasua narica has a higher than expected rmax (111% of predicted; Table 10). The life history feature that enhances Nasua narica's reproductive potential, and increases rmax beyond expected, is its large litter size. In this species females live in bands. Each year just before their young are born these bands break up, and each female seeks out a den for herself and [Pg 20] her litter. Once the young are able to leave the den (approximately five weeks), bands reform. In this situation, females not only care for their own young but also for those of other females in the band (Kaufmann, 1962:157-159, 1982, 1987; Russell, 1983). This social structure may contribute to this species' ability to produce large litters and in this way increase its reproductive potential.
Nasua narica.—This species is one of the largest members of the procyonid family (Table 7), and it has traits that should limit its ability to reproduce: a lower than expected Ḣb (Table 7), a relatively low-quality diet (Kaufmann, 1962:182-198; Table 9), and delayed first reproduction (Table 10). Despite this, Nasua narica has a higher than expected rmax (111% of predicted; Table 10). The life history trait that enhances Nasua narica's reproductive capacity and boosts rmax beyond predictions is its large litter size. In this species, females live in groups. Each year just before their young are born, these groups break up, and each female finds a den for herself and her litter. Once the young are able to leave the den (around five weeks), the groups come back together. In this scenario, females not only look after their own young but also help care for the young of other females in the group (Kaufmann, 1962:157-159, 1982, 1987; Russell, 1983). This social structure may help this species produce large litters and thereby improve its reproductive potential.
| Species | Body mass (g) | a | b | n | rmax | rmaxe[a] | rmaxr[b] | References |
|---|---|---|---|---|---|---|---|---|
| Procyon lotor | 4940 | 0.83 | 2.25 | 16 | 1.34 | 0.53 | 2.52 | Dunn and Chapman (1983); Eisenberg (1981:489); Kaufmann (1987); Lotze and Anderson (1979); Nowak and Paradiso (1983:981); Sanderson(1987); Stains (1956:28-31); This study |
| Bassariscus astutus | 900 | 0.83 | 1.50 | 14 | 1.02 | 0.82 | 1.24 | Kaufmann (1982, 1987); Nowak and Paradiso(1983:979, 980); Poglayen-Neuwall and Poglayen-Neuwall (1980); Poglayen-Neuwall and Toweill (1988); Russell (1983) |
| Nasua narica | 3900 | 2.50 | 2.25 | 14 | 0.62 | 0.56 | 1.11 | Kaufmann (1982, 1987); Nowak and Paradiso (1983:983); Sanderson (1983) |
| Nasua nasua | 3850 | Chevillard-Hugot et al. (1980) | ||||||
| Procyon cancrivorus | 1160 | 0.83 | 1.50 | 15 | 1.02[c] | 0.77 | 1.32 | Crandall (1964:312); Poglayen-Neuwall (1987) |
| 1.75 | 0.65[c] | 0.84 | ||||||
| Potos flavus | 2490 | 1.75 | 0.50 | 12 | 0.30 | 0.63 | 0.48 | Ford and Hoffmann (1988); Nowak and Paradiso (1983:984) |
| Bassaricyon gabbii | 1600 | 1.75 | 0.50 | 15 | 0.32 | 0.71 | 0.45 | Eisenberg (1981:489); Nowak and Paradiso (1983:985) |
[b] Regression of rmax on body mass (m). Assume rmax = 1.02 for Procyon cancrivorus: rmax = 0.00005·m + 0.623; R = 0.19; R2 = 0.03; Regression of rmaxr (Table 10) on Hbr (Table 7); assume Nasua nasua has the same rmaxr as Nasua narica: rmaxr = 3.35·Hbr - 1.11; R = 0.93; R2 = 0.86.
[c] Estimate based on females reproducing in their first (a = 0.83) or second (a = 1.75) year.
Nasua nasua.—Unfortunately, there is not enough reproductive data to allow calculation of rmax for Nasua nasua (Table 10), therefore, it is not possible to compare the reproductive potential of this South American coati with its North American relative, Nasua narica. Given its low Ḣb and relatively low-quality diet of fruit and terrestrial invertebrates (Table 9), however, rmax of Nasua nasua may be very similar to that of Nasua narica.
Nasua nasua.—Unfortunately, there isn't enough reproductive data to calculate rmax for Nasua nasua (Table 10), so we can't compare the reproductive potential of this South American coati to its North American counterpart, Nasua narica. However, given its low Ḣb and relatively low-quality diet of fruit and ground-dwelling invertebrates (Table 9), rmax for Nasua nasua may be quite similar to that of Nasua narica.
Procyon cancrivorus.—The age of first female reproduction for Procyon cancrivorus has not been reported. However, if one assumes females can reproduce in their first year, rmax for Procyon cancrivorus would be 1.02 (132% of expected; Table 10). If, on the other hand, first female reproduction is delayed until the second year, rmax would be 0.65 (84% of predicted; Table 10). Procyon cancrivorus has a low Ḣb, reduced litter size, and small body mass. Its low Ḣb may limit litter size, but as with Bassariscus astutus, the quality of its diet (a high percentage of small vertebrates; Table 9) and its small body size may make it possible for females to reproduce in their first year and thus increase the species' reproductive potential. This reasoning would argue that Procyon cancrivorus probably enjoys higher, rather than lower, than expected rmax.
Procyon cancrivorus.—The age at which female Procyon cancrivorus first reproduces hasn't been reported. However, if we assume that females can reproduce in their first year, rmax for Procyon cancrivorus would be 1.02 (132% of what was expected; Table 10). If, on the other hand, the first reproduction is delayed until the second year, rmax would be 0.65 (84% of what was predicted; Table 10). Procyon cancrivorus has a low Ḣb, reduced litter size, and small body mass. Its low Ḣb might limit the litter size, but similar to Bassariscus astutus, the quality of its diet (a high percentage of small vertebrates; Table 9) and its small body size may enable females to reproduce in their first year, thereby increasing the species' reproductive potential. This suggests that Procyon cancrivorus likely has a higher, not lower, than expected rmax.
Potos flavus.—In addition to a low Ḣb, this species possesses other characteristics that limit its reproductive potential: low-quality diet, delayed reproduction, and birth of a single young each year. Because there does not appear to be any other feature of its life history that can counteract the influence of these factors, rmax in Potos flavus has evolved to be only 48% of expected (0.30; Table 10). Its close relative, the olingo, Bassaricyon gabbii, appears to share the same condition (Table 10).
Potos flavus.—Along with a low Ḣb, this species has other traits that limit its reproductive potential: a low-quality diet, delayed reproduction, and typically only having one young each year. Since there doesn’t seem to be any other aspect of its life history that can offset these factors, rmax in Potos flavus has developed to be just 48% of what’s expected (0.30; Table 10). Its close relative, the olingo, Bassaricyon gabbii, appears to experience the same situation (Table 10).
Summary.—This brief survey illustrates that, with the exception of Potos flavus, procyonids tend to have values of rmax that are higher than those predicted for them on the basis of mass (Table 10). Regression analysis indicates that, within the family, body mass accounts for only a small amount (3%) of the variation in rmax, whereas the positive slope of the correlation between rmaxr and Hbr (R = 0.93) suggests that low metabolism has a limiting effect on rmax (see Table 10, footnote b). The implication here is that low Ḣb would be associated with a lower rate of biosynthesis, a slower growth rate, and a longer generation time. Procyonids with low Ḣb but higher than expected rmax must possess other traits that serve to offset the effects of low metabolism. Our survey indicates that the following features compensate for low Ḣb and help increase rmax: (1) a high-quality diet may make biosynthesis and growth more efficient, thus optimizing the time element associated [Pg 21] with each of these processes; (2) larger litter sizes and cooperation in care of the young may increase survivorship in spite of a slower growth rate; and (3) an early age of first reproduction, a long reproductive life span, and moderate-size litters (two to four young) may in the long run add as many individuals to the population as a shortened generation time. Our survey also suggests that, at the other extreme, factors such as a low-quality diet, reduced litter size, absence of cooperative care of the young, delayed age of first reproduction, and shortened reproductive life span all serve to decrease rmax. Thus, it is obvious that diet, litter size, social structure, reproductive strategy, and reproductive life span can operate synergistically with Ḣb to magnify its influence on rmax (as with Procyon lotor and Potos flavus), or they can function in opposition to Ḣb to change the direction of its influence on rmax (as with Bassariscus astutus, Procyon cancrivorus, Nasua narica, and perhaps Nasua nasua).
Summary.—This brief survey shows that, except for Potos flavus, procyonids generally have rmax values that are higher than what would be predicted based on their mass (Table 10). Regression analysis reveals that, within the family, body mass explains only a small portion (3%) of the variation in rmax, while the positive slope of the correlation between rmax and Hbr (R = 0.93) suggests that low metabolism restricts rmax (see Table 10, footnote b). This implies that low Ḣb is linked to a lower biosynthesis rate, slower growth, and longer generation times. Procyonids with low Ḣb but higher-than-expected rmax must have other traits that mitigate the effects of low metabolism. Our survey suggests that the following characteristics help compensate for low Ḣb and boost rmax: (1) a high-quality diet may enhance the efficiency of biosynthesis and growth, optimizing the duration of each process; (2) larger litter sizes and cooperative care of the young might improve survival rates despite a slower growth rate; and (3) an early age of first reproduction, a long reproductive lifespan, and moderately sized litters (two to four young) may ultimately contribute as many individuals to the population as a shorter generation time. Our survey also indicates that at the other end, factors like a low-quality diet, smaller litter sizes, lack of cooperative care of the young, delayed age of first reproduction, and shorter reproductive lifespan all lead to a decrease in rmax. Therefore, it is clear that diet, litter size, social structure, reproductive strategy, and reproductive lifespan can work together with Ḣb to amplify its influence on rmax (as seen in Procyon lotor and Potos flavus), or they can act against Ḣb to alter its impact on rmax (as observed in Bassariscus astutus, Procyon cancrivorus, Nasua narica, and possibly Nasua nasua).
Procyon lotor.—The evolution of a higher Ḣb (Tables 7, 8) may have been the physiological cornerstone that enabled Procyon lotor to break out of the mold being exploited by other procyonids and to generalize its use of habitats and climates. Once this basic physiological change was in place, selection for appropriate alterations in thermal conductance, capacity for evaporative cooling, diversity of diet, and energy storage would have provided this species with the suite of adaptations needed to extend its distribution into other habitats and climates. Support for this concept follows from the fact that high levels of Ḣb are associated with (1) cold-hardiness in mammals that live in cold-temperate and arctic climates (Scholander et al., 1950c; Irving et al., 1955; Irving, 1972:115, 116; Shield, 1972; Vogel, 1980; Golightly and Ohmart, 1983); (2) the ability to utilize a wide variety of food resources and to occupy a large number of different environments and habitats (McNab, 1980a); and (3) a high intrinsic rate of natural increase (McNab, 1980a; Hennemann, 1983; Lillegraven et al., 1987; Nicoll and Thompson, 1987; Thompson, 1987).
Procyon lotor.—The evolution of a higher Ḣb (Tables 7, 8) may have been the physiological key that allowed Procyon lotor to break free from the limitations faced by other procyonids and to adapt its use of various habitats and climates. Once this essential physiological change occurred, the selection for appropriate modifications in thermal conductance, the ability to cool through evaporation, dietary diversity, and energy storage would have equipped this species with the necessary adaptations to expand its range into different environments and climates. Support for this idea comes from the observation that high levels of Ḣb are associated with (1) cold-hardiness in mammals that inhabit cold-temperate and arctic regions (Scholander et al., 1950c; Irving et al., 1955; Irving, 1972:115, 116; Shield, 1972; Vogel, 1980; Golightly and Ohmart, 1983); (2) the capability to utilize a wide range of food resources and to thrive in many different environments and habitats (McNab, 1980a); and (3) a high intrinsic rate of natural increase (McNab, 1980a; Hennemann, 1983; Lillegraven et al., 1987; Nicoll and Thompson, 1987; Thompson, 1987).
Other Procyonids.—Other procyonids (Potos flavus, Procyon cancrivorus, Nasua narica, and Nasua nasua) have lower than predicted Ḣb's (Table 7), a characteristic that is considered to be an energy-saving adaptation for those that live in relatively stable tropical and subtropical habitats (Müller and Kulzer, 1977; Chevillard-Hugot et al., 1980; Müller and Rost, 1983). However, Bassariscus astutus is found in tropical, subtropical, and temperate climates. This species is found from tropical Mexico to temperate regions of the western United States (Kaufmann, 1982, 1987; Nowak and Paradiso, 1983:979). In the northern part of its distribution, Bassariscus astutus lives in habitats that are unstable (arid regions), that are low in productivity, and that characteristically have marked seasonal changes in temperature. Its lower than predicted Ḣb could be an important water-conserving adaptation at times when temperatures are high (McNab and Morrison, 1963; McNab, 1966; MacMillen and Lee, 1970; Noll-Banholzer, 1979) and an important energy-conserving mechanism when cold weather may limit food availability and hunting time (Scholander et al., 1950c; Wang et al., 1973). As will be seen later, Bassariscus astutus is unique among procyonids with lower than predicted Ḣb's in that it also has a lower than predicted Cmw (Table 7). This allows it to use less energy than expected for thermoregulation at low temperatures. Another species with a similar set of adaptations (lower than predicted Ḣb and Cmw) is the arctic hare, Lepus arcticus (Wang et al., 1973), which lives in one of the coldest and least-productive regions on earth. Wang et al. (1973) suggest that this combination of adaptations allows Lepus arcticus to better match its energy requirements to the low productivity of its environment. A similar relationship may hold for Bassariscus astutus, particularly in colder arid portions of its distribution, and may be the reason that it, but not other procyonids with low Ḣb's, has been able to inhabit temperate climates.
Other raccoons.—Other procyonids (Potos flavus, Procyon cancrivorus, Nasua narica, and Nasua nasua) have lower than expected Ḣb's (Table 7), which is seen as an energy-saving adaptation for those living in relatively stable tropical and subtropical environments (Müller and Kulzer, 1977; Chevillard-Hugot et al., 1980; Müller and Rost, 1983). However, Bassariscus astutus can be found in tropical, subtropical, and temperate climates. This species ranges from tropical Mexico to the temperate areas of the western United States (Kaufmann, 1982, 1987; Nowak and Paradiso, 1983:979). In the northern part of its range, Bassariscus astutus inhabits unstable (arid) regions that have low productivity and significant seasonal temperature fluctuations. Its lower than expected Ḣb might be an important water-conserving adaptation during high temperatures (McNab and Morrison, 1963; McNab, 1966; MacMillen and Lee, 1970; Noll-Banholzer, 1979) and a crucial energy-conserving mechanism when cold weather may limit food availability and hunting time (Scholander et al., 1950c; Wang et al., 1973). As will be discussed later, Bassariscus astutus stands out among procyonids with lower than expected Ḣb's because it also has a lower than predicted Cmw (Table 7). This trait enables it to use less energy than anticipated for thermoregulation in low temperatures. Another species with a similar adaptation pattern (lower than expected Ḣb and Cmw) is the arctic hare, Lepus arcticus (Wang et al., 1973), which resides in one of the coldest and least productive regions on earth. Wang et al. (1973) propose that this combination of adaptations allows Lepus arcticus to align its energy needs better with the low productivity of its habitat. A comparable relationship may exist for Bassariscus astutus, especially in the colder arid areas it inhabits, which may explain why it, unlike other procyonids with low Ḣb's, has managed to occupy temperate climates.
Thermal conductance is a measure of the ease with which heat is passively transferred to or from a body through its tissues and pelt. Within Tn, a mammal is able to vary its thermal conductance over a wide range of values by changing heat transfer characteristics of both of these layers. Minimum thermal conductance occurs when total heat transfer through these layers is reduced to its lowest possible rate. This minimum value, which is the reciprocal of maximum resistance, occurs, theoretically, but not always practically (see McNab, 1988b), at the animal's Tlc and is best estimated under standard conditions in a metabolism chamber (McNab, 1980b; Aschoff, 1981). Minimum thermal conductance scales to body mass (McNab and Morrison, 1963; Herreid and Kessel, 1967; McNab, 1970, 1979b; Bradley and Deavers, 1980; Aschoff, 1981). Therefore, to make comparisons between species of various sizes, we scaled out body mass by expressing Cmw as the ratio of measured to predicted values (Cmwr; Table 7). These ratios were used to make comparisons of heat-transfer characteristics between species that occupy different habitats or climates.
Thermal conductance measures how easily heat is passively transferred to or from a body through its tissues and skin. Within Tn, a mammal can adjust its thermal conductance across a wide range by changing the heat transfer properties of both layers. Minimum thermal conductance occurs when total heat transfer through these layers is reduced to the lowest possible rate. This minimum value, which is the opposite of maximum resistance, theoretically occurs at the animal's Tlc, but not always in practice (see McNab, 1988b). It's best estimated under standard conditions in a metabolism chamber (McNab, 1980b; Aschoff, 1981). Minimum thermal conductance scales with body mass (McNab and Morrison, 1963; Herreid and Kessel, 1967; McNab, 1970, 1979b; Bradley and Deavers, 1980; Aschoff, 1981). Therefore, to compare species of different sizes, we adjusted for body mass by expressing Cmw as the ratio of measured to predicted values (Cmwr; Table 7). These ratios were used to compare heat transfer characteristics among species that live in different habitats or climates.
In summer, Tlc's of male and female Procyon lotor (Figure 2) were very similar to those of other procyonids (22°C-26°C; Table 7). In winter, Tlc of both sexes shifted downward to 11°C (Figure 3). This seasonal shift in Tlc occurred as the result of a seasonal change in minimum thermal conductance (Table 3). For many northern mammals, a seasonal change in thermal conductance is partly mediated via cyclic changes in the insulative quality of their pelt (Scholander et al., 1950a; Irving et al., 1955; Hart, 1956, 1957; Irving, 1972:165).
In summer, the Tlc of male and female Procyon lotor (Figure 2) was very similar to that of other procyonids (22°C-26°C; Table 7). In winter, the Tlc for both sexes dropped to 11°C (Figure 3). This seasonal change in Tlc happened due to a seasonal shift in minimum thermal conductance (Table 3). For many mammals in northern regions, the seasonal variation in thermal conductance is partly influenced by changes in the insulative properties of their fur (Scholander et al., 1950a; Irving et al., 1955; Hart, 1956, 1957; Irving, 1972:165).
[Pg 22] Procyon lotor begins to shed its heavy winter coat about the time its young are born. Molt progresses through summer and by late August the new coat is complete (Stuewer, 1942). During its summer molt, Procyon lotor's Cmw increased by about 49% over the value for female raccoons in winter (Table 3). In summer, therefore, it had the highest mass specific Cmw of those procyonids considered (Cmwr = 1.77 and 1.79; Table 7). An increase in thermal conductance facilitates passive heat loss for temperate and arctic species, and this serves as an important thermoregulatory adaptation during warm summer months (Scholander et al., 1950c; Irving et al., 1955; Hart, 1956, 1957; Irving, 1972:165). This adaptation is particularly important to those temperate- and arctic-zone species (including raccoons) whose Ḣb's do not decrease during summer (Irving et al., 1955). From August on, the fur of Procyon lotor becomes increasingly longer and heavier, with peak, or prime, condition occurring in late fall and early winter (Stuewer, 1942). Minimum conductance of our captive raccoons was lowest in winter (Cmwr = 1.15) when their pelts were in prime condition (Tables 3, 7). Because "primeness" of raccoon pelts varies geographically, thicker pelts being associated with colder climates (Goldman, 1950:21; Whitney and Underwood, 1952:24-41), the degree of seasonal change in Cmw must also vary geographically.
[Pg 22] Procyon lotor starts shedding its thick winter coat around the time its young are born. The molting process continues through the summer, and by late August, the new coat is fully grown (Stuewer, 1942). During the summer molt, Procyon lotor's Cmw increased by about 49% compared to the value for female raccoons in winter (Table 3). Therefore, in the summer, it had the highest mass-specific Cmw among the procyonids studied (Cmwr = 1.77 and 1.79; Table 7). An increase in thermal conductance helps with passive heat loss for species from temperate and arctic regions, making it a key thermoregulation strategy during the warm summer months (Scholander et al., 1950c; Irving et al., 1955; Hart, 1956, 1957; Irving, 1972:165). This adaptation is especially vital for temperate and arctic species (like raccoons) whose Ḣb's don’t drop in the summer (Irving et al., 1955). Starting in August, the fur of Procyon lotor grows longer and heavier, reaching its peak or prime condition in late fall and early winter (Stuewer, 1942). The minimum conductance of our captive raccoons was lowest in winter (Cmwr = 1.15) when their fur was in prime condition (Tables 3, 7). Since the "primeness" of raccoon pelts differs geographically, with thicker pelts linked to colder climates (Goldman, 1950:21; Whitney and Underwood, 1952:24-41), the level of seasonal change in Cmw must also vary by location.
The only other procyonid for which a seasonal molt has been described is Bassariscus astutus. Molt in this species extends from late summer to late fall (Toweill and Toweill, 1978). How molt effects thermal conductance in Bassariscus astutus is not known because metabolic data for this species (Table 7) apparently were collected only when their pelts were in prime condition (Chevalier, 1985).
The only other procyonid known to have a seasonal molt is Bassariscus astutus. In this species, molting occurs from late summer to late fall (Toweill and Toweill, 1978). It's unclear how molting affects thermal conductance in Bassariscus astutus because metabolic data for this species (Table 7) seem to have been gathered only when their fur was in prime condition (Chevalier, 1985).
Goldman (1950:20) reports that Procyon cancrivorus does not have a seasonal molt. Like other tropical procyonids, Procyon cancrivorus lives in an environment that has the following characteristics: high even temperatures throughout the year (1°C-13°C difference in monthly mean temperature), a greater range in temperature between day and night than in mean monthly temperature throughout the year, uniform lengths of day and night, seasonal variation in rainfall, and lowest temperatures during the rainy season(s) (Kendeigh, 1961:340). In such a stable environment there would be no advantage to a sharply defined seasonal molt cycle that could place an animal in thermoregulatory jeopardy by increasing its thermal conductance. This would be particularly true for animals like tropical procyonids that have lower than predicted Ḣb's but that maintain typical eutherian body temperatures (Table 7). Consequently, molt in all tropical procyonids may either be prolonged or continuous. This is a feature of their biology that needs to be examined in more detail.
Goldman (1950:20) reports that Procyon cancrivorus does not experience a seasonal molt. Like other tropical procyonids, Procyon cancrivorus inhabits an environment with these characteristics: consistently high temperatures all year round (a 1°C-13°C difference in monthly average temperatures), a greater temperature variation between day and night compared to the average monthly temperature, equal lengths of day and night, seasonal changes in rainfall, and the lowest temperatures during the rainy season(s) (Kendeigh, 1961:340). In such a stable environment, there would be no benefit to having a sharply defined seasonal molt cycle, as it could put the animal at risk for thermal regulation by increasing its thermal conductance. This is especially relevant for tropical procyonids, which have lower than expected Ḣb's but maintain typical eutherian body temperatures (Table 7). Therefore, molting in all tropical procyonids may either be extended or continuous. This is a biological aspect that requires further investigation.
Procyon lotor versus Tropical Procyonids.—Cmwr for Procyon lotor in winter was 1.15, which is similar to the values for Potos flavus and Procyon cancrivorus, 1.02 and 1.25, respectively (Table 7). These two tropical species, therefore, have Cmw's that are similar on a mass specific basis to the value for Procyon lotor in winter. However, at their Tlc's, the thermal gradient sustained by these tropical animals is only about 11°C, whereas for Procyon lotor in winter it was 26.5°C. Examination of Eq. 4 with respect to these thermal gradients suggests that tropical procyonids achieve such low Cmw's by virtue of their lower than predicted Ḣb's rather than by having pelts that are exceptionally good insulators. In fact, the insulation afforded by the pelts of these tropical procyonids is about the same as that of the 50 g arctic lemming, Dicrostonyx groenlandicus rubricatus, whose coat has an insulative value that is about half that of the hare, Lepus americanus, red fox, Vulpes fulva alascensis, and pine martin, Martes americana, animals comparable in size to these procyonids (Scholander et al., 1950a). Therefore, pelts of these tropical procyonids do not have the same insulative value as the prime winter coat of Procyon lotor.
Procyon lotor versus Tropical Procyonids.—Cmwr for Procyon lotor in winter was 1.15, which is similar to the values for Potos flavus and Procyon cancrivorus, which are 1.02 and 1.25, respectively (Table 7). These two tropical species, then, have Cmw's that are comparable on a mass-specific basis to the value for Procyon lotor in winter. However, at their Tlc's, the thermal gradient maintained by these tropical animals is only about 11°C, while for Procyon lotor in winter it was 26.5°C. Analyzing Eq. 4 regarding these thermal gradients indicates that tropical procyonids achieve such low Cmw's due to their lower than expected Ḣb's rather than having pelts that are exceptionally good insulators. In fact, the insulation provided by the pelts of these tropical procyonids is pretty much the same as that of the 50 g arctic lemming, Dicrostonyx groenlandicus rubricatus, whose coat has an insulative value that is about half that of the hare, Lepus americanus, the red fox, Vulpes fulva alascensis, and the pine marten, Martes americana, which are animals comparable in size to these procyonids (Scholander et al., 1950a). Therefore, the pelts of these tropical procyonids do not have the same insulative value as the prime winter coat of Procyon lotor.
Nasua narica and Nasua nasua have tropical and subtropical distributions and they are the only procyonids that are diurnal (Kaufmann, 1962:103-105, 1982, 1987). Because they are active during the day they experience a more extreme thermal environment (higher Ta's and solar radiation) than their nocturnal cousins. Values of Cmwr for Nasua narica (1.45 and 1.55) and Nasua nasua (1.24 and 1.65) are higher than those for Procyon cancrivorus or Potos flavus (Table 7). Thus, these coatis have higher mass specific Cmw's than their nocturnal tropical cousins. A high Cmw reduces the cost of thermoregulation in hot environments because it increases an animal's ability to lose excess heat passively. The higher Cmw's of these coatis serve as an adaptation that contributes to the success of their diurnal life style as well as their ability to expand their habitat use to areas with less thermal stability, such as oak and pine woodlands and deserts.
Nasua narica and Nasua nasua are found in tropical and subtropical areas, and they're the only procyonids that are active during the day (Kaufmann, 1962:103-105, 1982, 1987). Since they are active in daylight, they face a more extreme thermal environment (higher Ta's and solar radiation) compared to their nocturnal relatives. The values of Cmwr for Nasua narica (1.45 and 1.55) and Nasua nasua (1.24 and 1.65) are higher than those for Procyon cancrivorus or Potos flavus (Table 7). This means these coatis have a higher mass-specific Cmw than their nocturnal tropical cousins. A higher Cmw reduces the cost of thermoregulation in hot environments because it enhances an animal's ability to lose excess heat passively. The increased Cmw of these coatis is an adaptation that supports their successful diurnal lifestyle and enables them to use habitats with less thermal stability, such as oak and pine woodlands and deserts.
Bassariscus astutus.—This species has the lowest mass specific Cmw of these procyonids (Cmwr = 0.85; Table 7), which indicates that its pelt has a greater insulative value than the coats of Potos flavus, Procyon cancrivorus, Nasua nasua, or Nasua narica. This, coupled with a lower than predicted Ḣb, allows Bassariscus astutus to maintain Tb with less energy expenditure than is possible for any other procyonid of comparable size; and this combination of adaptations provides Bassariscus astutus with a distinct energy advantage in environments that have low productivity (Wang et al., 1973). The evolution of a pelt that provides better insulation must be considered an, important contributing factor for the spread of this species into desert regions of the western United States.
Bassariscus astutus.—This species has the lowest mass-specific Cmw among these procyonids (Cmwr = 0.85; Table 7), which means its fur is more insulating than that of Potos flavus, Procyon cancrivorus, Nasua nasua, or Nasua narica. This, along with a lower than expected Ḣb, allows Bassariscus astutus to maintain Tb with less energy use than any other similarly sized procyonid; this combination of traits gives Bassariscus astutus a clear energy advantage in less productive environments (Wang et al., 1973). The development of fur that offers better insulation is a significant factor in the species' expansion into desert areas of the western United States.
Thermoregulation.—At temperatures below a mammal's Tn, heat loss exceeds Ḣb. To maintain Tb under these [Pg 23] conditions, metabolic rate must be increased (Eq. 4). Procyon lotor in summer during its annual molt (Table 5; Figure 2), Bassariscus astutus (Chevalier, 1985), Nasua nasua (Chevillard-Hugot et al., 1980; Mugaas et al., in prep.), Nasua narica (Scholander et al., 1950b; Mugaas et al., in prep.), and Potos flavus (Müller and Kulzer, 1977; Müller and Rost, 1983) all are able to elevate their metabolic rates by 130% above basal when they are exposed to Ta = 0°C. Procyon cancrivorus responds to 0°C with an increase in metabolic rate of 257% above basal (Scholander et al., 1950b). All animals listed have about the same Tlc and Tb, so the temperature differential producing this response is about the same for each species. Metabolic ability to defend body temperature against low ambient temperatures, therefore, is well developed in these procyonids. Such large increases in metabolic rate are energetically expensive, and if these animals were routinely exposed to Ta = 0°C, it would be difficult for them to acquire enough food each day to maintain endothermy. Raccoons in winter pelage, however, need only elevate their metabolic rate by 47% above basal to maintain endothermy at Ta = 0°C (Table 5; Figure 3). Each year at the completion of its molt, the raccoon's highly insulative pelt is renewed. This lowers their Tlc by 9°C to 15°C below that measured for them in summer (Figure 3) and decreases their cost of thermoregulation at low temperatures. The increased insulative capacity of their pelt is one of the primary adaptations that has allowed Procyon lotor to extend its distribution into cold climates.
Body temperature control.—At temperatures below a mammal's Tn, heat loss is greater than Ḣb. To keep Tb stable under these conditions, the metabolic rate must be raised (Eq. 4). Procyon lotor in summer during its annual molt (Table 5; Figure 2), Bassariscus astutus (Chevalier, 1985), Nasua nasua (Chevillard-Hugot et al., 1980; Mugaas et al., in prep.), Nasua narica (Scholander et al., 1950b; Mugaas et al., in prep.), and Potos flavus (Müller and Kulzer, 1977; Müller and Rost, 1983) can all boost their metabolic rates by 130% above baseline when exposed to Ta = 0°C. Procyon cancrivorus increases its metabolic rate by 257% above baseline in response to 0°C (Scholander et al., 1950b). All the animals listed have similar Tlc and Tb, so the temperature difference triggering this response is about the same for each species. Therefore, their metabolic ability to regulate body temperature against low surrounding temperatures is well developed. Such significant increases in metabolic rate require a lot of energy, and if these animals were frequently exposed to Ta = 0°C, they would struggle to find enough food daily to sustain endothermy. However, raccoons in winter fur only need to raise their metabolic rate by 47% above baseline to maintain endothermy at Ta = 0°C (Table 5; Figure 3). Each year, when their molt ends, the raccoon's highly insulating fur is renewed. This lowers their Tlc by 9°C to 15°C compared to summer measurements (Figure 3) and reduces their thermoregulation costs at low temperatures. The increased insulation of their fur is one of the main adaptations that has enabled Procyon lotor to expand its range into colder climates.
Stored Fat.—Cyclic fattening is an integral and important part of a raccoon's annual cycle (Mugaas and Seidensticker, ms); however, it has not been reported for other procyonids. During winter in parts of the United States and Canada, raccoons are confined to their dens for variable periods of time (days to months) depending on the severity of the weather (Stuewer, 1943:223-225; Whitney and Underwood, 1952:108-116; Sharp and Sharp, 1956; Mech et al., 1968; Schneider et al., 1971). During this confinement, they do not hibernate but rather enter a state of "dormancy" and become inactive. While dormant they remain endothermic (Tb > 35°C; Thorkelson, 1972:87-90) and derive most of their energy requirement from fat reserves accumulated during fall. The rate at which fat stores are consumed during winter dormancy depends on the thermoregulatory requirement imposed on them by local weather conditions, the insulative quality of their pelt, and any advantage they may gain by seeking shelter in a den.
Body Fat. — Cyclic fattening is a crucial and significant part of a raccoon's yearly cycle (Mugaas and Seidensticker, ms); however, it hasn't been reported in other procyonids. During winter in some areas of the United States and Canada, raccoons are stuck in their dens for varying amounts of time (from days to months) depending on how severe the weather is (Stuewer, 1943:223-225; Whitney and Underwood, 1952:108-116; Sharp and Sharp, 1956; Mech et al., 1968; Schneider et al., 1971). While they are confined, they don't hibernate but go into a state of "dormancy" and become inactive. During dormancy, they remain warm-blooded (Tb > 35°C; Thorkelson, 1972:87-90) and get most of their energy from fat reserves built up in the fall. The rate at which they use up their fat stores during winter dormancy relies on the thermoregulation required by local weather conditions, the insulating quality of their fur, and any benefits they might gain from finding shelter in a den.
Heat transfer between an animal and its environment is a function of the interaction of its body temperature and thermal conductance with various environmental variables (air temperature, wind speed, vapor pressure, and thermal radiation). When a raccoon is outside its den, its thermal conductance (Cmw) is the only barrier to heat transfer with the external environment. However, when it enters a tree den, a raccoon imposes two other thermal barriers between itself and the external environment: (1) conductance of the air space between its fur and the den's walls (Ca) and (2) conductance of the den's walls (Cd; Thorkelson, 1972:59-63; Thorkelson and Maxwell, 1974). Thorkelson and Maxwell (1974) modeled heat transfer of a simulated raccoon (a water-filled aluminum cylinder equipped with a heater and covered with a raccoon pelt) in a closed tree den. In their system, 65% of resistance to heat flux was attributable to the pelt, whereas the remainder (35%) was due to Ca and Cd. Because resistance is the inverse of conductance, and resistances for the raccoon and its den are arranged in series, we can estimate total conductance (Ct) of this system with Eq. 7.
Heat transfer between an animal and its environment depends on how its body temperature and thermal conductance interact with various environmental factors like air temperature, wind speed, vapor pressure, and thermal radiation. When a raccoon is outside its den, its thermal conductance (Cmw) is the only thing that limits heat transfer with the external environment. However, when it goes into a tree den, a raccoon creates two additional thermal barriers between itself and the outside: (1) the conductance of the air space between its fur and the walls of the den (Ca), and (2) the conductance of the den's walls (Cd; Thorkelson, 1972:59-63; Thorkelson and Maxwell, 1974). Thorkelson and Maxwell (1974) simulated heat transfer in a raccoon (represented by a water-filled aluminum cylinder with a heater, covered by a raccoon pelt) within a closed tree den. In their setup, 65% of the resistance to heat flow was due to the pelt, while the remaining 35% came from Ca and Cd. Since resistance is the opposite of conductance, and the resistances for the raccoon and its den are arranged in series, we can estimate the total conductance (Ct) of this system with Eq. 7.
| 1/Ct = 1/Cmw + 1/Ca + 1/Cd | Eq. 7 |
Minimum thermal conductance Cmw for raccoons in winter was 0.0172 mL O2·g-1·h-1·°C-1 (Table 3). Based on Thorkelson and Maxwell's (1974) model we let 1/Cmw = 0.65(1/Ct) = 1/0.0172 mL O2·g-1·h-1·°C-1, and 1/Ca + 1/Cd = 0.35(1/Ct). Substituting these values into Eq. 7 and solving for Ct yields 0.0112 mL O2·g-1·h-1·°C-1, a value that is 35% lower than that of the animal alone. Substituting this value and the value for basal metabolism of winter raccoons (0.47 mL O2·g-1·h-1; Table 7) into Eq. 4 and solving for (Tb - Ta) yields a new temperature differential of 42°C. Therefore, by using tree dens, raccoons in north central Virginia, with Tb = 37°C (Figure 7), could effectively reduce their Tlc from 11°C to -5°C and markedly reduce their metabolic cost of thermoregulation.
Minimum thermal conductance Cmw for raccoons in winter was 0.0172 mL O2·g-1·h-1·°C-1 (Table 3). Based on Thorkelson and Maxwell's (1974) model we let 1/Cmw = 0.65(1/Ct) = 1/0.0172 mL O2·g-1·h-1·°C-1, and 1/Ca + 1/Cd = 0.35(1/Ct). Substituting these values into Eq. 7 and solving for Ct gives 0.0112 mL O2·g-1·h-1·°C-1, which is 35% lower than that of the animal alone. Plugging this value and the basal metabolism of winter raccoons (0.47 mL O2·g-1·h-1; Table 7) into Eq. 4 and solving for (Tb - Ta) gives a new temperature difference of 42°C. So, by using tree dens, raccoons in north central Virginia, with Tb = 37°C (Figure 7), could effectively lower their Tlc from 11°C to -5°C and significantly cut down on their metabolic cost of thermoregulation.
Given prevailing winter temperatures in north central Virginia (see "Materials and Methods"), adult raccoons in that area should be able to sustain endothermy most of the time they are in their dens by simply maintaining Ḣb. Depending on the mass of their stored fat, they could remain in their dens for several weeks without eating (Mugaas and Seidensticker, ms). The thermal advantage of a den could be further enhanced during colder temperatures if two or more raccoons occupied it at the same time and huddled together, and/or if these animals could reduce Cmw even more by lowering Tb and cooling their extremities. Although we do not have any data to verify the second mechanism, there are many accounts in natural history literature that document raccoons occupying dens together (Lotze and Anderson, 1979). This habit could be particularly important for the young of the year and may be one reason why they often continue to den with their mothers during winter (Lotze and Anderson, 1979; Seidensticker et al., 1988). Raccoons that live in colder climates, such as Minnesota, undoubtedly obtain the same advantage from a den as Virginia animals, but because of their greater body mass, longer fur, and potentially lower Cmw, Tlc of a Minnesota raccoon in a den could be even lower than what we calculated for Virginia raccoons. Therefore, when they are in their dens, raccoons living in very cold climates also may be able to maintain homeothermy with a basal level of metabolism.[Pg 24]
Given the winter temperatures in north central Virginia (see "Materials and Methods"), adult raccoons in that area should be able to maintain their body heat most of the time while in their dens by simply keeping their body temperature stable. Depending on how much fat they've stored, they could stay in their dens for several weeks without eating (Mugaas and Seidensticker, ms). The benefit of a den could increase in colder weather if two or more raccoons used it together and huddled up, or if they could lower their body temperature further and cool their limbs. Although we lack data to support the second mechanism, many accounts in natural history literature mention raccoons sharing dens (Lotze and Anderson, 1979). This behavior could be especially important for the young ones and might explain why they often stay with their mothers in the winter (Lotze and Anderson, 1979; Seidensticker et al., 1988). Raccoons living in colder regions, like Minnesota, likely enjoy the same benefits from a den as those in Virginia, but due to their larger body size, thicker fur, and potentially lower heat loss, the temperature in a Minnesota raccoon's den could be even lower than what we've calculated for Virginia raccoons. Thus, when in their dens, raccoons in extremely cold climates may also be able to maintain their body heat with just a basic level of metabolism.[Pg 24]
In hot environments mammals depend on behavior to minimize their thermal load (escape to shaded or cooler microclimates, use posture and orientation to wind and sun, restrict activity, become nocturnal, etc.) and on evaporative water loss to rid themselves of excess heat. With regard to evaporative heat loss, Calder and King (1974:326) arbitrarily subdivided the response to various Ta's as follows: "(1) cool temperatures at which water loss should be minimized, both to reduce heat loss and as an adaptation to terrestriality; (2) an intermediate temperature range wherein evaporation is gradually increased as dry heat losses are proportionately reduced with smaller thermal gradients; and (3) warm to hot temperatures at which evaporation must be actively increased to dispose of metabolic and exogenous heat loads." Some mammals are able to thermoregulate very well at high ambient temperatures via panting or sweating, whereas others have a very limited capacity. Hence, there is no general approach to calculating evaporative water loss under these conditions (Campbell, 1977:85). However, the ratio of evaporative heat lost to metabolic heat produced can be used to quantify a species' capacity for evaporative cooling and to make comparisons between species.
In hot environments, mammals rely on their behavior to reduce their heat load (like seeking shade or cooler spots, adjusting their posture in relation to the wind and sun, limiting their activity, or becoming nocturnal) and on losing water through evaporation to get rid of extra heat. Regarding evaporative heat loss, Calder and King (1974:326) divided the response to different air temperatures (Ta) into three categories: "(1) cool temperatures where water loss should be minimized to lower heat loss and adapt to living on land; (2) an intermediate temperature range where evaporation gradually increases as dry heat losses are reduced with smaller temperature differences; and (3) warm to hot temperatures where evaporation has to be actively increased to eliminate both metabolic and external heat." Some mammals manage to regulate their body temperature effectively in high ambient temperatures by panting or sweating, while others have a limited ability to do so. Therefore, there isn't a standard way to calculate evaporative water loss in these situations (Campbell, 1977:85). However, the ratio of evaporative heat lost to metabolic heat produced can be used to measure a species' ability to cool down through evaporation and to compare different species.
Potos flavus.—This species lives in Neotropical forests of Central and South America. It is nocturnal, arboreal in habit, and appears to be the most heat-sensitive of these procyonids. Its Tuc is at 30°C to 33°C (Table 7; Müller and Kulzer, 1977; Müller and Rost, 1983). It begins to pant at about 30°C, but its efforts at evaporative cooling are very ineffective. At 33°C Potos flavus can dissipate 33% of its metabolic heat via evaporative water loss, but at 35°C the efficiency of this mechanism falls to 20% (Müller and Rost, 1983). Consequently, when exposed to Ta's above 33°C, any kind of excitement causes its Tb to rise rapidly in an uncontrolled manner (Müller and Kulzer, 1977; Müller and Rost, 1983). These animals rely on their nocturnal and arboreal habits to keep them out of situations that could lead to hyperthermia (Müller and Kulzer, 1977; Müller and Rost, 1983).
Potos flavus.—This species is found in the tropical forests of Central and South America. It is active at night, lives in trees, and seems to be the most sensitive to heat among these raccoons. Its Tuc is between 30°C and 33°C (Table 7; Müller and Kulzer, 1977; Müller and Rost, 1983). It starts panting at around 30°C, but its ability to cool itself through evaporation is quite ineffective. At 33°C, Potos flavus can lose 33% of its metabolic heat through evaporative water loss, but at 35°C, that efficiency drops to 20% (Müller and Rost, 1983). As a result, when exposed to Ta's above 33°C, any excitement causes its body temperature (Tb) to increase quickly and uncontrollably (Müller and Kulzer, 1977; Müller and Rost, 1983). These animals depend on their nocturnal and tree-dwelling habits to avoid situations that could lead to overheating (Müller and Kulzer, 1977; Müller and Rost, 1983).
Nasua nasua and Nasua narica.—Nasua nasua is abundant in tropical and subtropical South America, whereas Nasua narica occupies the same climates in North America from southern Arizona and New Mexico south through Panama and on into Colombia and Ecuador (Hall and Kelson, 1959:892; Ewer, 1973:391, 392; Poglayen-Neuwall, 1975). Both coatis are diurnal and forage primarily on the ground (Kaufmann, 1962:185-188, 1987; Poglayen-Neuwall, 1975; Nowak and Paradiso, 1983:982), consequently they are exposed to a more severe thermal environment while active (higher Ta's and solar radiation) than are nocturnal procyonids. Both coatis are more heat-tolerant than Potos flavus; their Tuc's are higher (33°C-35°C; Table 7), they can tolerate Ta's of 35°C without raising their Tb's (Chevillard-Hugot et al., 1980; Mugaas et al., in prep.), and they have a greater capacity for evaporative cooling than Potos flavus (Mugaas et al., in prep.). The greater heat tolerance of these coatis is compatible with their diurnal habits and widespread distribution in a variety of forest habitats in both tropical and subtropical areas of the western hemisphere.
Nasua nasua and Nasua narica.—Nasua nasua is common in tropical and subtropical regions of South America, while Nasua narica is found in similar climates in North America, from southern Arizona and New Mexico, south through Panama and into Colombia and Ecuador (Hall and Kelson, 1959:892; Ewer, 1973:391, 392; Poglayen-Neuwall, 1975). Both coatis are active during the day and primarily search for food on the ground (Kaufmann, 1962:185-188, 1987; Poglayen-Neuwall, 1975; Nowak and Paradiso, 1983:982), which means they face more extreme heat and sunlight while active compared to nocturnal procyonids. Both coatis can handle heat better than Potos flavus; their upper critical temperatures (Tuc) are higher (33°C-35°C; Table 7), they can manage ambient temperatures (Ta) of 35°C without increasing their body temperatures (Tb) (Chevillard-Hugot et al., 1980; Mugaas et al., in prep.), and they have a better ability to cool themselves through evaporation than Potos flavus (Mugaas et al., in prep.). This greater heat tolerance aligns with their daytime activity and wide distribution in various forest habitats in both tropical and subtropical areas of the western hemisphere.
Bassariscus astutus.—In addition to living in Neotropical forests of Mexico, Bassariscus astutus also flourishes in hot arid climates, and it has extended its range much farther north than Nasua narica (Hall and Kelson, 1959:881,892; Poglayen-Neuwall, 1975; Kaufmann, 1982). Its Tuc is higher (35.5°C; Table 7) than that of Potos flavus, but it is comparable to those of Nasua nasua and Nasua narica. Its capacity for evaporative cooling is well developed; at 40°C Bassariscus astutus is able to dissipate 100% of its resting metabolic heat via evaporative water loss, and at 45°C it is able to dissipate 172% (Chevalier, 1985). In spite of its great capacity for evaporative cooling, this species is nocturnal, a habit that, along with its low Ḣb, should allow it to keep thermoregulatory water requirements to a minimum.
Bassariscus astutus.—Besides inhabiting the Neotropical forests of Mexico, Bassariscus astutus also thrives in hot, dry climates, and it has expanded its range much further north than Nasua narica (Hall and Kelson, 1959:881,892; Poglayen-Neuwall, 1975; Kaufmann, 1982). Its Tuc is higher (35.5°C; Table 7) than that of Potos flavus, but it is similar to those of Nasua nasua and Nasua narica. Its ability for evaporative cooling is highly developed; at 40°C, Bassariscus astutus can lose 100% of its resting metabolic heat through evaporative water loss, and at 45°C it can lose 172% (Chevalier, 1985). Despite its excellent ability for evaporative cooling, this species is nocturnal, a behavior that, along with its low Ḣb, should enable it to keep its thermoregulatory water needs to a minimum.
Procyon lotor.—Our data suggested that Tuc for Procyon lotor in winter was comparable to that for Bassariscus astutus (35°C), and that in summer it was even higher. When exposed to temperatures near the upper end of its Tn, Procyon lotor increased the gradient for passive heat loss with a controlled rise in Tb (Figure 6). In summer its capacity for passive heat loss was enhanced by the molt of its heavy winter fur. Procyon lotor's capacity for evaporative cooling also appeared to be well developed, although our animals were not heated to the point that evaporative cooling was fully expressed (Figures 4, 5). However, Procyon lotor is nocturnal, and this may allow it to eliminate, or at least reduce, the need for evaporative cooling, even in hot climates. Thus, Procyon lotor appears to be well equipped physiologically and behaviorally to cope with thermal demands of hot environments in its distribution.
Procyon lotor.—Our data indicated that Tuc for Procyon lotor in winter was similar to that for Bassariscus astutus (35°C), and that in summer it was even higher. When exposed to temperatures close to the upper limit of its Tn, Procyon lotor increased the gradient for passive heat loss with a controlled rise in Tb (Figure 6). In summer, its ability to lose heat passively was enhanced by shedding its heavy winter fur. Procyon lotor's capacity for evaporative cooling also seemed to be well developed, although our animals were not heated to the point that evaporative cooling was fully active (Figures 4, 5). However, Procyon lotor is nocturnal, which may help it minimize, or at least lessen, the need for evaporative cooling, even in hot climates. Therefore, Procyon lotor seems well adapted both physiologically and behaviorally to handle the thermal challenges of hot environments in its range.
Procyon cancrivorus.—Unfortunately, data for the crab-eating raccoon are not complete enough at high temperatures to include it in this survey.
Procyon cancrivorus.—Unfortunately, the data for the crab-eating raccoon are not comprehensive enough at high temperatures to include it in this survey.
Summary.—This comparison demonstrates that capacity for evaporative cooling, tolerance of an elevated Tb to enhance passive heat loss, and behavioral avoidance of thermal stress are the primary methods used by procyonids to thermoregulate at high temperatures. Procyon lotor and Bassariscus astutus, whose distributions extend into temperate regions, have developed these abilities to a greater extent than other procyonids. Potos flavus, whose distribution is confined to lowland tropical forests, has the least ability in this regard. Nasua nasua and Nasua narica appear to have thermoregulatory abilities that are intermediate to those of Bassariscus astutus and Potos flavus. This suggests that ancestral procyonids [Pg 25] may have had poor to modest ability to thermoregulate at high temperatures, a condition that would have limited their ability to leave the thermal stability afforded by tropical forests. Dispersal into temperate climates, therefore, required not only increased cold tolerance but also selective enhancement of those mechanisms used in thermoregulation at high temperatures.
Summary.—This comparison shows that the ability to cool through evaporation, endure higher body temperatures to boost passive heat loss, and avoid thermal stress behaviorally are the main strategies procyonids use to regulate their body temperature in hot environments. Procyon lotor and Bassariscus astutus, which are found in temperate areas, have developed these abilities more than other procyonids. Potos flavus, which is only found in lowland tropical forests, has the least capability in this aspect. Nasua nasua and Nasua narica seem to have thermoregulatory abilities that are in between those of Bassariscus astutus and Potos flavus. This indicates that early procyonids [Pg 25] might have had limited to modest abilities to regulate their temperature in hot conditions, which would have restricted their capability to move away from the thermal stability of tropical forests. Dispersing into temperate climates, therefore, required not just improved cold tolerance but also a selective enhancement of the mechanisms used for temperature regulation in high heat situations.
| Species | Tropics | Subtropics | Mild[a] temperate | Cold[b] temperate | ||
|---|---|---|---|---|---|---|
| Procyon lotor | + | + | + | + | ||
| Bassariscus astutus | + | + | + | |||
| Nasua nasua | + | + | ||||
| Nasua narica | + | + | ||||
| Procyon cancrivorus | + | + | ||||
| Potos flavus | + | |||||
[b] Extends northward from the 10°C isotherm for average annual temperature in the United States.
In Table 11, procyonid species are arranged in descending order with respect to the number of major climates that are included in their geographic distributions (Hall and Kelson, 1959:878-897; Poglayen-Neuwall, 1975; Kortlucke and Ramirez-Pulido, 1982; Nowak and Paradiso, 1983:977-985). Composite scores ranged from a high of 1.47 for Procyon lotor to a low of 0.39 for Potos flavus, whereas Nasua nasua, Nasua narica, Procyon cancrivorus, and Bassariscus astutus had intermediate values ranging from 0.64 to 0.79 (Table 12). Figure 8 demonstrates that there is a direct relationship between the number of climates these species occupy and their composite scores. Regression analysis (Y = 2.68·X + 0.24; where Y is number of climates, and X is composite score) demonstrates a high degree of correlation between these variables (R = 0.94) and indicates that 89% of the variance in distribution can be explained by composite scores. The various combinations of adaptations expressed by these species do, therefore, play a role in delimiting their climatic (latitudinal) distributions.
In Table 11, procyonid species are listed in order from the most to the least based on the number of major climates within their geographic ranges (Hall and Kelson, 1959:878-897; Poglayen-Neuwall, 1975; Kortlucke and Ramirez-Pulido, 1982; Nowak and Paradiso, 1983:977-985). Composite scores varied from a high of 1.47 for Procyon lotor to a low of 0.39 for Potos flavus, while Nasua nasua, Nasua narica, Procyon cancrivorus, and Bassariscus astutus showed intermediate scores ranging from 0.64 to 0.79 (Table 12). Figure 8 reveals a direct relationship between the number of climates these species inhabit and their composite scores. Regression analysis (Y = 2.68·X + 0.24; where Y represents the number of climates, and X represents the composite score) demonstrates a strong correlation between these variables (R = 0.94) and indicates that 89% of the variation in distribution can be explained by composite scores. The different combinations of adaptations shown by these species do, therefore, influence their climatic (latitudinal) distributions.
Procyon lotor's normalized scores were higher in all categories than those of other procyonids. Procyon lotor, therefore, possesses those traits that have allowed it to become the premier climate generalist of the procyonid family. As an adaptive unit, these traits provide Procyon lotor with the physiological and behavioral flexibility required to take full advantage of a wide range of climates and habitats, and its distribution verifies that it has done so. Even so, it is probably not fair to assume that this species represents a perfect physiological match with climate over its entire distribution. Procyon lotor is, in many respects, still a forest-dwelling species, and its ability to expand its distribution into other habitats such as prairie and desert may well be due, in part, to its use of behavior to take advantage of favorable microclimates in otherwise hostile environments (Bartholomew, 1958, 1987). This feature of Procyon lotor's biology needs to be further examined.
Procyon lotor had higher normalized scores in all categories compared to other procyonids. Therefore, Procyon lotor has the traits that have enabled it to become the leading climate generalist in the procyonid family. As an adaptive unit, these traits give Procyon lotor the physiological and behavioral flexibility needed to thrive in a variety of climates and habitats, and its distribution confirms that it has successfully done so. However, it’s likely not accurate to assume that this species perfectly matches with the climate throughout its entire range. Procyon lotor is, in many ways, still primarily a forest-dwelling species, and its ability to spread into other environments like prairies and deserts may partially stem from its use of behavior to exploit favorable microclimates in otherwise challenging areas (Bartholomew, 1958, 1987). This aspect of Procyon lotor's biology needs to be studied further.
| Species | Normalized scores | Composite[a] score | |||
|---|---|---|---|---|---|
| Hbr/Cmwr | Ddr | rmaxr | |||
| Procyon lotor | 0.95 | 0.95 | 2.52 | 1.47 | |
| Bassariscus astutus | 0.80 | 0.33 | 1.24 | 0.79 | |
| Nasua nasua | 0.48 | 0.33 | 1.11[b] | 0.64 | |
| Nasua nasua | 0.48 | 0.33 | 1.11[b] | 0.64 | |
| Nasua narica | 0.40 | 0.53 | 1.11 | 0.68 | |
| Procyon cancrivorus | 0.55 | 0.33 | 1.32 | 0.73 | |
| Potos flavus | 0.60 | 0.11 | 0.48 | 0.39 | |
[b] Value calculated for Nasua narica (Table 10) and used with the assumption that it must be similar to the value for Nasua nasua.
All five species with low Ḣb's have composite scores less than 1.0 (Table 12; Figure 8). Four of these five, Nasua nasua, Nasua narica, Procyon cancrivorus, and Potos flavus, have Hbr/Cmwr ratios that are 0.6 or less, which indicates they are the least cold-tolerant procyonids (McNab, 1966). These four species also are confined to either tropic, or tropic and subtropic climates (Table 11). This suggests that these species share a common thermoregulatory adaptation that represents a specialization to these climates. Attendant with this adaptation, however, is a high cost of thermoregulation at [Pg 26] temperatures below their Tlc, and this must be an important factor in limiting their distributions to tropic and subtropic climates. Differences in their distributions within these climates, therefore, must hinge more on differences in their Ddr and rmaxr values than on differences in their Hbr/Cmwr ratios. This is supported by the fact that Potos flavus, which has the lowest Ddr and rmaxr values, is confined to a single climate, whereas Nasua nasua, Nasua narica, and Procyon cancrivorus each possess larger Ddr and rmaxr values and are found in two climates. Thus, Potos flavus, with its highly specialized diet and low reproductive potential, is the most ecologically specialized of these procyonids, and its distribution is limited to the single climate that can provide its requirements. Nasua nasua, Nasua narica, and Procyon cancrivorus are less specialized and thus show more ecological flexibility in their distributions.
All five species with low Ḣb's have composite scores of less than 1.0 (Table 12; Figure 8). Four of these species, Nasua nasua, Nasua narica, Procyon cancrivorus, and Potos flavus, have Hbr/Cmwr ratios that are 0.6 or less, indicating that they are the least cold-tolerant procyonids (McNab, 1966). These four species are also restricted to either tropical or both tropical and subtropical climates (Table 11). This suggests that these species share a common thermoregulatory adaptation that represents a specialization for these climates. However, this adaptation comes with a high thermoregulation cost at temperatures below their Tlc, which is likely a key factor limiting their distribution to tropical and subtropical climates. Therefore, differences in their distribution within these climates are likely more related to variations in their Ddr and rmaxr values rather than differences in their Hbr/Cmwr ratios. This is supported by the fact that Potos flavus, which has the lowest Ddr and rmaxr values, is confined to a single climate, while Nasua nasua, Nasua narica, and Procyon cancrivorus each have larger Ddr and rmaxr values and are found in two climates. Thus, Potos flavus, with its highly specialized diet and low reproductive potential, is the most ecologically specialized of these procyonids, and its distribution is limited to the specific climate that meets its needs. Nasua nasua, Nasua narica, and Procyon cancrivorus are less specialized, allowing them to demonstrate greater ecological flexibility in their distributions.

Bassariscus astutus, the other species with low Ḣb, is found in three climates, which indicates that it has greater ecological flexibility than Nasua nasua, Nasua narica, or Procyon cancrivorus. Ddr and rmaxr are comparable for these four species (Table 12). This suggests that the greater ecological flexibility of Bassariscus astutus is derived largely from its greater cold tolerance. Bassariscus astutus has a more insulative pelt than these other procyonids (Cmwr = 0.85; Table 7), so its Hbr/Cmwr ratio is higher (0.80; Table 12). This, and its greater capacity for evaporative cooling (Chevalier, 1985), allows Bassariscus astutus to take advantage of a wider range of thermal environments than these other species. However, even with its higher Hbr/Cmwr ratio, the composite score for Bassariscus astutus is not much different than those for Nasua nasua, Nasua narica, and Procyon cancrivorus (Table 12). Consequently, Bassariscus astutus is found in more climates than would be predicted for it on the basis of its composite score (Figure 8). This suggests that either the Hbr/Cmwr ratio carries greater weight in determining distribution than is reflected in this analysis, or as has been described for some other species (Bartholomew, 1958, 1987), Bassariscus astutus may extend its distribution farther than expected via use of its behavior. In either case, for procyonids with low Ḣb, Bassariscus astutus represents the pinnacle of adaptation for climate generalization.
Bassariscus astutus, the other species with low Ḣb, is found in three climates, which shows that it has more ecological flexibility than Nasua nasua, Nasua narica, or Procyon cancrivorus. Ddr and rmaxr are similar for these four species (Table 12). This indicates that the greater ecological flexibility of Bassariscus astutus comes mainly from its better cold tolerance. Bassariscus astutus has a more insulative fur than these other procyonids (Cmwr = 0.85; Table 7), so its Hbr/Cmwr ratio is higher (0.80; Table 12). This, along with its improved ability for evaporative cooling (Chevalier, 1985), allows Bassariscus astutus to thrive in a wider range of thermal environments than these other species. However, even with its higher Hbr/Cmwr ratio, the overall score for Bassariscus astutus is not much different from those for Nasua nasua, Nasua narica, and Procyon cancrivorus (Table 12). As a result, Bassariscus astutus is found in more climates than would be expected based on its overall score (Figure 8). This suggests that either the Hbr/Cmwr ratio plays a bigger role in determining distribution than this analysis shows, or as has been observed for some other species (Bartholomew, 1958, 1987), Bassariscus astutus may extend its range further than anticipated through its behavior. In either case, for procyonids with low Ḣb, Bassariscus astutus represents the height of adaptation for climate generalization.
A radiation of frugivorous and omnivorous Procyoninae (Table 1) occurred in the middle and late Miocene of North America. It included origins of such terrestrial genera as Cyonasua, Nasua, and Procyon (Webb, 1985b). The earliest procyonid genus to find its way to South America was Cyonasua, an omnivorous carnivore that presumably split, along with its sister genus Arctonasua, from a common North American ancestor (Baskin, 1982; Webb, 1985b). Cyonasua, about the size of present-day raccoons, was adapted to a wide range of habitats and was probably comparable to modern raccoons with respect to the breadth of its feeding habits (Webb, 1985b; Marshall, 1988). Because North American Arctonasua was about the same size as Cyonasua (Webb, 1985b) and shared a number of characters with it (Baskin, 1982), we speculate that it also may have had similar habits and occupied similar climates and habitats. Bassariscus, another member of Procyoninae, had an even earlier origin in tropical North America (Webb, 1985b). The origin of the small arboreal forms Potos and Bassaricyon (subfamily Potosinae) is obscure but is thought to have occurred in the rainforests of Central America (Webb, 1985b). What were the metabolic capabilities of these early procyonids? We do not know, but for several million years, from middle to late Miocene, procyonids lived in tropical and subtropical forests of Central and North America (Webb, 1985b; Marshall, 1988). Then, in the Pleistocene, several modern forms crossed the Panamanian land bridge into similar habitats and climates in South America; but none of them appear to have spread far enough northward to have crossed the Bering land bridge.
A variety of fruit-eating and omnivorous Procyoninae (Table 1) emerged in the middle and late Miocene in North America. This included the beginnings of terrestrial genera like Cyonasua, Nasua, and Procyon (Webb, 1985b). The first procyonid genus to reach South America was Cyonasua, an omnivorous carnivore that likely split from its sister genus Arctonasua, which shared a common North American ancestor (Baskin, 1982; Webb, 1985b). Cyonasua, roughly the size of today’s raccoons, adapted to a variety of habitats and was probably similar to modern raccoons in terms of its diverse diet (Webb, 1985b; Marshall, 1988). Since North American Arctonasua was about the same size as Cyonasua (Webb, 1985b) and had several traits in common with it (Baskin, 1982), we think it also likely had similar habits and lived in comparable climates and habitats. Bassariscus, another member of Procyoninae, originated even earlier in tropical North America (Webb, 1985b). The origins of the small tree-dwelling forms Potos and Bassaricyon (subfamily Potosinae) are unclear but are believed to have taken place in the rainforests of Central America (Webb, 1985b). What were the metabolic abilities of these early procyonids? We don’t know, but for several million years, from the middle to late Miocene, procyonids thrived in the tropical and subtropical forests of Central and North America (Webb, 1985b; Marshall, 1988). Then, during the Pleistocene, several modern species crossed the Panamanian land bridge into similar environments and climates in South America; however, none of them seem to have traveled far enough north to cross the Bering land bridge.
Several million years exposure to a tropical environment, with its continuous high temperatures and modest range of thermal extremes, would have favored selection of metabolic and thermoregulatory traits that would minimize energy requirements: a lower than predicted basal metabolic rate, a prolonged or continuous molt resulting in very little annual change in minimum thermal conductance, and a modest capacity for evaporative cooling. In addition, we would expect selection to have favored a diverse diet, good reproductive [Pg 27] potential, and behavioral flexibility to utilize a variety of habitats within these climates. Our analysis has shown that such characteristics are the norm for extant members of this family living in tropical and subtropical climates, and we speculate that these traits also were common to early procyonids and served to restrict them to these climates. Our speculation is supported by the fact that their known fossil history from the Miocene is confined to geographic areas that had tropical and subtropical climates.
Several million years in a tropical environment, with its constantly high temperatures and limited temperature extremes, would have led to the selection of metabolic and thermoregulation traits that reduce energy needs: a lower than expected basal metabolic rate, a long or continuous molt resulting in minimal annual changes in thermal conductance, and a moderate ability for evaporative cooling. Furthermore, we would expect that natural selection would have favored a varied diet, strong reproductive potential, and behavioral flexibility to adapt to different habitats in these climates. Our analysis indicates that these traits are typical for current members of this family living in tropical and subtropical regions, and we speculate that these characteristics were also common among early procyonids, helping to limit them to these climates. Our speculation is backed by the fact that their known fossil history from the Miocene is restricted to geographic areas with tropical and subtropical climates.
Later on, during Pleistocene glaciations, tropical and subtropical forests shrank, savannas expanded, and temperate climate was pushed toward equatorial regions. The opposite occurred during interglacial periods (Raven and Axelrod, 1975; Webb, 1977, 1978; Marshall, 1988). Consequently, mid-latitudes experienced alternating periods of temperate and tropical, or at least subtropical, climate change. Selection of characteristics that would have adapted a species with low Ḣb to temperate as well as tropic or subtropic climates could have occurred in mid-latitudes at the temperate edge of these tropical advances and retreats. Our analysis indicates that, for this purpose, selection would have favored lower than predicted thermal conductance, seasonal molt, increased capacity for evaporative cooling, increased tolerance of elevated Tb, increased flexibility of thermoregulatory behavior, food habits that provided for year-round access to a high-quality diet in all three climates, and a higher than predicted rmax.
Later on, during the Pleistocene ice ages, tropical and subtropical forests decreased, savannas increased, and temperate climates moved closer to the equator. The reverse happened during interglacial periods (Raven and Axelrod, 1975; Webb, 1977, 1978; Marshall, 1988). As a result, mid-latitudes went through alternating periods of temperate and tropical, or at least subtropical, climate changes. Selection of traits that would have helped a species with low Ḣb adapt to both temperate and tropical or subtropical climates could have happened in mid-latitudes along the temperate edges of these tropical shifts. Our analysis suggests that, for this reason, selection would have favored lower than expected thermal conductance, seasonal molting, a greater ability for evaporative cooling, increased tolerance for higher Tb, greater flexibility in thermoregulatory behavior, dietary habits that ensured year-round access to a high-quality diet across all three climates, and a higher than expected rmax.
Bassariscus astutus is the only species with low Ḣb that has all these characteristics, and it is the only one of them that has added temperate climate to its distribution (Table 11). This suggests that Bassariscus astutus is a species that evolved away from the norm for procyonids with low Ḣb, toward characteristics that allowed it to become more of a climate generalist. Potos flavus, with its dietary specialization, low tolerance to high temperatures, and arboreal mode of existence, has become a highly specialized species totally dependent on tropical forests for its survival. As such, it also represents a species that has evolved away from the procyonid norm and portrays the extreme in climate specialization. Olingos, Bassaricyon gabbii (Table 1), may be similar to Potos flavus in this respect (see also Table 10). This suggests that of the extant procyonids, Nasua nasua, Nasua narica, and Procyon cancrivorus have retained metabolic and behavioral characteristics that are closest to those of their Miocene ancestors.
Bassariscus astutus is the only species with low Ḣb that possesses all these traits, and it’s the only one that has expanded its range to include temperate climates (Table 11). This indicates that Bassariscus astutus is a species that has evolved away from the typical traits of procyonids with low Ḣb, moving towards characteristics that enable it to thrive in a wider range of climates. On the other hand, Potos flavus, with its specialized diet, low tolerance for high temperatures, and tree-dwelling lifestyle, has become highly specialized and totally reliant on tropical forests for survival. Therefore, it also exemplifies a species that has evolved away from the procyonid norm and represents an extreme case of climate specialization. Olingos, Bassaricyon gabbii (Table 1), may share similarities with Potos flavus in this regard (see also Table 10). This suggests that among the living procyonids, Nasua nasua, Nasua narica, and Procyon cancrivorus have preserved metabolic and behavioral traits that align most closely with those of their Miocene ancestors.
Between the time that Cyonasua appeared and the Panamanian land bridge was established in the upper Pliocene (4 to 5 million years ago), northern climates continued their gradual cooling. This, along with ongoing elevation of the continents and continuous modification of their mountain ranges, served to shrink the tropical forest and create pockets of climatic instability within it and on its edges (Darlington, 1963:578-596; Marshall, 1988). In areas of instability, selection would have favored traits that provided for a broader range of thermal tolerance: higher Ḣb, improved insulative quality of pelt, a more sharply defined molt cycle, improved capacity for evaporative cooling, greater Dd, and higher rmax. Consequently, by the upper Pliocene, two metabolically distinct groups of procyonids could have been established: those species with low Ḣb living in climatically stable forests and those with higher Ḣb living in unstable tropical, subtropical, and perhaps temperate climates.
Between the time that Cyonasua appeared and the Panamanian land bridge formed in the upper Pliocene (4 to 5 million years ago), northern climates continued to gradually cool. This, along with the ongoing uplift of the continents and continuous changes in their mountain ranges, caused the tropical forest to shrink and created pockets of climatic instability within it and on its edges (Darlington, 1963:578-596; Marshall, 1988). In unstable areas, natural selection would have favored traits that allowed for a broader range of thermal tolerance: higher Ḣb, improved insulation quality of fur, a more clearly defined molt cycle, better capacity for evaporative cooling, greater Dd, and higher rmax. As a result, by the upper Pliocene, two metabolically distinct groups of procyonids could have emerged: those species with low Ḣb living in climatically stable forests and those with higher Ḣb inhabiting unstable tropical, subtropical, and possibly temperate climates.
Procyon lotor is the only extant procyonid with high Ḣb. Procyon cancrivorus is its congeneric counterpart in Central and South America (Table 1), and the two species are sympatric in Panama and Costa Rica. However, in terms of its metabolism, thermal conductance, molt, diversity of diet, rmax, and climatic distribution, Procyon cancrivorus shares more in common with other procyonids than it does with Procyon lotor (Tables 7, 11, 12; Figure 8). This suggests that metabolically Procyon lotor portrays a divergent line of this genus that arose as the result of a series of mutations that gave rise to different metabolic characteristics. This view is in keeping with a recent phylogenetic analysis of this family that shows the genus Procyon to be highly derived (Decker and Wozencraft, 1991). Consequently, it would be instructive and would add to our knowledge of the evolution of climatic adaptation to know more about the genetic relatedness of these two species as well as their historical relationship.
Procyon lotor is the only living procyonid with high Ḣb. Procyon cancrivorus is its closely related counterpart in Central and South America (Table 1), and both species coexist in Panama and Costa Rica. However, in terms of metabolism, thermal conductance, molting, diet diversity, rmax, and climate distribution, Procyon cancrivorus has more in common with other procyonids than with Procyon lotor (Tables 7, 11, 12; Figure 8). This indicates that metabolically, Procyon lotor represents a distinct line within this genus that resulted from a series of mutations leading to different metabolic traits. This perspective aligns with a recent phylogenetic analysis of this family, revealing that the genus Procyon is highly specialized (Decker and Wozencraft, 1991). Thus, it would be valuable to learn more about the genetic connections between these two species and their historical relationship, which would enhance our understanding of the evolution of climate adaptation.
Genus Procyon appears in the fossil record (Hemphillian and Blancan ages; Baskin, 1982) prior to Pleistocene glaciations. During the Pleistocene, there were four different glacial advances and retreats in a relatively short time period (the first appearing little more than a million years ago; Darlington, 1963:578-596; Webb, 1985a; Marshall, 1988). Glacial retreats created pulses of time during which subtropic and temperate climates advanced toward the poles into areas with large seasonal differences in light/dark cycles, whereas glacial advances pushed these climates southward into areas having smaller seasonal differences in light/dark cycles (Raven and Axelrod, 1975; Webb, 1977, 1978; Marshall, 1988). Those members of the genus Procyon caught in these wide latitudinal fluctuations would have experienced conditions favorable to continued selection for characteristics conducive to physiologic adaptation to a wide range of climatic conditions. Procyon lotor is the only member of its genus to have survived this selective process, and as we have seen, it does possess traits that adapt it to a wide range of climatic conditions. Primary among these is its higher Ḣb, which provides it with advantages not shared with other procyonids (see earlier discussion). Three other adaptations also have had a profound influence on Procyon lotor's ability to generalize its use of climate: (1) the increased insulative quality of its pelt coupled with its sharply defined molt cycle, which allows for a large annual change in thermal conductance; (2) its annual cycle of fat storage; and (3) a diverse high-quality diet. The first two of these adaptations [Pg 28] required evolution of neuroendocrine pathways capable of responding to time-dependent environmental cues such as changing day length, changing temperature, etc. Such conditions would have been available as selective stimuli in high-latitude forests and savannas of interglacial periods. Procyon lotor's elevated basal metabolic rate would have increased its overall energy requirement, and it makes good intuitive sense, therefore, that evolution during the Pleistocene also would have favored selection of a diverse diet containing many items of high nutritive value.
Genus Procyon appears in the fossil record (Hemphillian and Blancan ages; Baskin, 1982) before the Pleistocene glaciations. During the Pleistocene, there were four different glacial advances and retreats in a relatively short time frame (the first one occurring just over a million years ago; Darlington, 1963:578-596; Webb, 1985a; Marshall, 1988). Glacial retreats created time periods where subtropical and temperate climates moved toward the poles into regions with significant seasonal differences in light/dark cycles, while glacial advances pushed these climates south into areas with smaller seasonal differences in light/dark cycles (Raven and Axelrod, 1975; Webb, 1977, 1978; Marshall, 1988). Members of the genus Procyon caught in these broad latitudinal shifts would have experienced conditions that favored the ongoing selection of traits that enable physiological adaptation to a variety of climatic conditions. Procyon lotor is the only surviving member of its genus, and as we have seen, it has traits that allow it to adapt to different climatic conditions. Chief among these is its higher Ḣb, which gives it advantages not shared with other procyonids (see earlier discussion). Three other adaptations have also significantly impacted Procyon lotor's ability to adapt to various climates: (1) the enhanced insulative quality of its fur combined with its distinct molt cycle, allowing for a significant annual change in thermal conductance; (2) its yearly fat storage cycle; and (3) a varied, high-quality diet. The first two adaptations [Pg 28] required the evolution of neuroendocrine pathways that could respond to time-dependent environmental signals like changing day length and temperature. Such situations would have been present as selective influences in the high-latitude forests and savannas of interglacial periods. Procyon lotor's raised basal metabolic rate would have increased its overall energy needs, and it makes sense that evolution during the Pleistocene would also have favored the selection of a varied diet containing many high-nutrition items.
Our analysis has illustrated that within Procyonidae there are two distinct modes of metabolic adaptation to climate. One is typified by those species with low Ḣb's (Bassariscus astutus, Nasua nasua, Nasua narica, Procyon cancrivorus, and Potos flavus), and the other by Procyon lotor with its higher Ḣb. Those with low Ḣb's have more restricted geographic distributions, and, with the exception of Bassariscus astutus, they are all confined to tropical and subtropical areas. The fossil history of this family indicates that it had its origins in tropical forests of North and Central America. This indicates that those procyonids whose distributions are still primarily restricted to tropical forests share many of the metabolic adaptations characteristic of their ancestors. We speculate, therefore, that ancestral procyonids had a lower than predicted Ḣb, a pelt with modest to poor insulative quality, good thermogenic ability but poor heat tolerance, modest to poor capacity for evaporative cooling, no well-defined molt cycle, no cyclic period of fattening, nocturnal habits, and a modestly diverse diet of high-enough quality to provide for an average reproductive potential. Although this pedigree contributed to the success of this family in tropical and subtropical forests, it limited the ability of its members to expand their distributions into cooler, less stable climates. Viewed in this perspective, Procyon lotor's high basal metabolic rate, extraordinarily diverse diet, well-defined cyclic changes in fat content and thermal conductance, high level of heat tolerance, high capacity for evaporative cooling, and high reproductive potential all stand out in sharp contrast to the condition described for other procyonids. This suggests that the North American raccoon represents culmination of a divergent evolutionary event that has given this species the ability to break out of the old procyonid mold and carry the family into new habitats and climates.
Our analysis has shown that within the Procyonidae family, there are two different ways of adapting metabolically to climate. One is represented by species with low basal metabolic rates (like Bassariscus astutus, Nasua nasua, Nasua narica, Procyon cancrivorus, and Potos flavus), while the other is represented by Procyon lotor, which has a higher metabolic rate. The species with low metabolic rates tend to have more limited geographic ranges, and with the exception of Bassariscus astutus, they are all found mainly in tropical and subtropical regions. The fossil record for this family indicates that it originated in the tropical forests of North and Central America. This suggests that the procyonids that still mainly inhabit tropical forests have many of the same metabolic adaptations as their ancestors. We therefore speculate that ancestral procyonids had a lower than expected metabolic rate, a coat with mediocre to poor insulation, good ability to generate heat but poor tolerance to heat, moderate to poor evaporative cooling capabilities, no clear molting cycle, no regular fattening period, nocturnal behavior, and a moderately varied diet of high enough quality to support average reproductive potential. While these traits helped the family thrive in tropical and subtropical forests, they limited their ability to spread into cooler, less stable climates. From this perspective, Procyon lotor's high basal metabolic rate, extremely varied diet, clearly defined cycles in fat levels and thermal conductivity, high heat tolerance, strong evaporative cooling ability, and high reproductive potential significantly contrast with those of other procyonids. This indicates that the North American raccoon represents the culmination of a divergent evolutionary event that has allowed this species to step outside the traditional procyonid mold and adapt to new habitats and climates.
| a | potential age of females first producing young |
| b | potential annual birth rate of female young |
| Ca | conductance of air |
| Cd | conductance of den walls |
| Cm | minimum thermal conductance |
| Cmd | minimum dry thermal conductance |
| Cmw | minimum wet thermal conductance |
| Cmwr | ratio of measured to predicted minimum wet thermal conductance |
| Ct | total conductance |
| Dd | diversity of diet |
| Ddr | ratio of food categories actually used by a species to the total number of food categories taken by all species tested |
| Ė | evaporative water loss |
| Ec | ratio of evaporative heat lost to metabolic heat produced |
| Ėeq | oxygen equivalent for heat lost by evaporation |
| Ḣb | basal metabolic rate |
| Ḣr | lowest resting metabolic rate at each temperature |
| Hbr | ratio of measured to predicted basal metabolic rate |
| m | mass of animal |
| mw | mass of water |
| n | potential age of females producing their final young |
| rmax | intrinsic rate of natural increase |
| rmaxe | expected intrinsic rate of natural increase |
| rmaxr | ratio of calculated to expected intrinsic rate of natural increase |
| RQ | respiratory quotient |
| Ta | chamber air temperature |
| Tb | body temperature |
| Tlc | lower critical temperature |
| Tn | thermoneutral zone |
| Tuc | upper critical temperature |
| t | time |
| .Va | rate of air flow through |
| .Ve | rate of air flow into metabolism chamber |
| α | active phase of the daily cycle |
| γ | heat equivalent of oxygen |
| λ | heat of vaporization of water |
| ρ | rest phase of the daily cycle |
Emphasis upon publication as a means of "diffusing knowledge" was expressed by the first Secretary of the Smithsonian. In his formal plan for the institution, Joseph Henry outlined a program that included the following statement: "It is proposed to publish a series of reports, giving an account of the new discoveries in science, and of the changes made from year to year in all branches of knowledge." This theme of basic research has been adhered to through the years by thousands of titles issued in series publications under the Smithsonian imprint, commencing with Smithsonian Contributions to Knowledge in 1848 and continuing with the following active series:
Emphasizing publication as a way to "spread knowledge" was highlighted by the first Secretary of the Smithsonian. In his official plan for the institution, Joseph Henry outlined a program that included the following statement: "We plan to publish a series of reports that will detail new discoveries in science and the changes made each year across all fields of knowledge." This focus on fundamental research has been maintained over the years with thousands of titles published in series under the Smithsonian imprint, starting with Smithsonian Contributions to Knowledge in 1848 and continuing with the following active series:
Smithsonian Contributions to Botany
Smithsonian Contributions to the Earth Sciences
Smithsonian Contributions to the Marine Sciences
Smithsonian Contributions to Paleobiology
Smithsonian Contributions to Zoology
Smithsonian Folklife Studies
Smithsonian Studies in Air and Space
Smithsonian Studies in History and Technology
In these series, the Institution publishes small papers and full-scale monographs that report the research and collections of its various museums and bureaux or of professional colleagues in the world of science and scholarship. The publications are distributed by mailing lists to libraries, universities, and similar institutions throughout the world.
In these series, the Institution publishes short papers and full-length monographs that showcase the research and collections of its various museums and offices or from professional colleagues in the fields of science and scholarship. The publications are sent out via mailing lists to libraries, universities, and similar institutions around the world.
Papers or monographs submitted for series publication are received by the Smithsonian Institution Press, subject to its own review for format and style, only through departments of the various Smithsonian museums or bureaux, where the manuscripts are given substantive review. Press requirements for manuscript and art preparation are outlined on the inside back cover.
Papers or monographs submitted for series publication are accepted by the Smithsonian Institution Press, which will review them for format and style, only through the departments of the various Smithsonian museums or bureaus, where the manuscripts undergo thorough review. The press's guidelines for preparing manuscripts and artwork are detailed on the inside back cover.
Secretary
Smithsonian Institution
With the exception of the typographical corrections listed below and some minor changes that may have been made in moving tables or illustrations so that they are rejoined, the text presented is that published in the original printed media.
With the exceptions of the typographical corrections listed below and a few minor adjustments made to reposition tables or illustrations for cohesiveness, the text provided is the same as that published in the original print media.
| Page ii, LOC Data | : | Instituion's | => | Institution's |
| Page 1, Introduction | : | linages | => | lineages |
| Page 4, The Atypical Procyonid | : | consumate | => | consummate |
| Page 21,Summary | : | Table 10, footnote f | => | Table 10, footnote b |
| Page 26, first paragraph | : | Nassua | => | Nasua |
| Page 31, Literature Cited | : | Incoporated | => | Incorporated |
| Page 34, Literature Cited | : | Gettleman | => | Gittleman |
Download ePUB
If you like this ebook, consider a donation!